Session Information
Session Time: 5:00PM-6:00PM
Background/Purpose: India carries one of the highest global burdens of tuberculosis (TB). Children on TNF inhibitor(TNFi) therapy face an increased risk of latent TB infection (LTBI) reactivation or progression to active TB. This study aimed to determine the prevalence of active TB and LTBI in children with rheumatological disorders before starting TNF inhibitors, and incidence of new infections during follow up using standard TB Screening protocol.
Methods: A Retrospective observational study was undertaken at a tertiary care hospital. All children who received anti TNF from 1st January 2015 to 15th November 2025 were included. The baseline demographics, rheumatological diagnosis and pre biologic screening for TB (Mantoux, QG, Chest Xray, Abdominal ultrasound and selectively Computed Tomography Chest +/- Abdomen) were recorded for all children to be started on TNFi. Rescreening for TB, if available, was also recorded for these children. Conversion rates for each TNFi and Mantoux–QG concordance/discordance were calculated.
Results: A total of 551 children underwent primary TB screening prior to initiation of TNF inhibitors, of whom 345 (62.6%) were males. Juvenile Idiopathic Arthritis (478/551, 86.8%) was the predominant diagnosis amongst which Enthesitis related arthritis predominated (305/478, 63.8%), followed by uveitis (6.17%) and vasculitis (2.7%). Etanercept (44.6%) and Adalimumab (44.2%) were the most frequently used biologics at baseline. LTBI was seen in 13.7%(76/551), and active TB in 1.27%(7/551) prior to commencing anti TNF treatment.
Re-screening was available in 190 children. The median duration of TNF Exposure was 2.9 years (Range: 0.04 years-13.76 years). Of these, 132 (69.1%) were males, and JIA remained predominant, with ERA still leading (120/169, 71%). Etanercept (44.7%) and Adalimumab (35.7%) continued to be the major biologics. New LTBI was detected in 15.2% (29/190), and active TB in 4.73% (9/190) during treatment despite normal primary screening. Adalimumab showed the maximum conversion rate (23.53% (16/68; 95% CI 14.09–35.38). Despite small subgroups, all TNFi agents showed measurable TB conversions.
Mantoux–QG discordance rose from 14.3% at baseline to 28% on rescreening, indicating declining agreement and highlighting that a single test cannot reliably exclude TB.
Conclusion: Despite initial screening, substantial new LTBI (29, 15.2%) and active TB(9, 4.73%) developed in children on TNFi therapy during follow-up, stressing the need for periodic re-screening in these children in high burden settings. Lower LTBI incidence than national estimates (40%) may reflect our patients’ relatively higher socioeconomic profile. Conversion Rates were maximum with Adalimumab in our cohort. Persistent Mantoux–QG discordance supports standardized, multimodal, and context-specific screening strategies in children with rheumatological disorders on TNFi.
Demographic Details of the children with rheumatological disorders at baseline and on rescreening 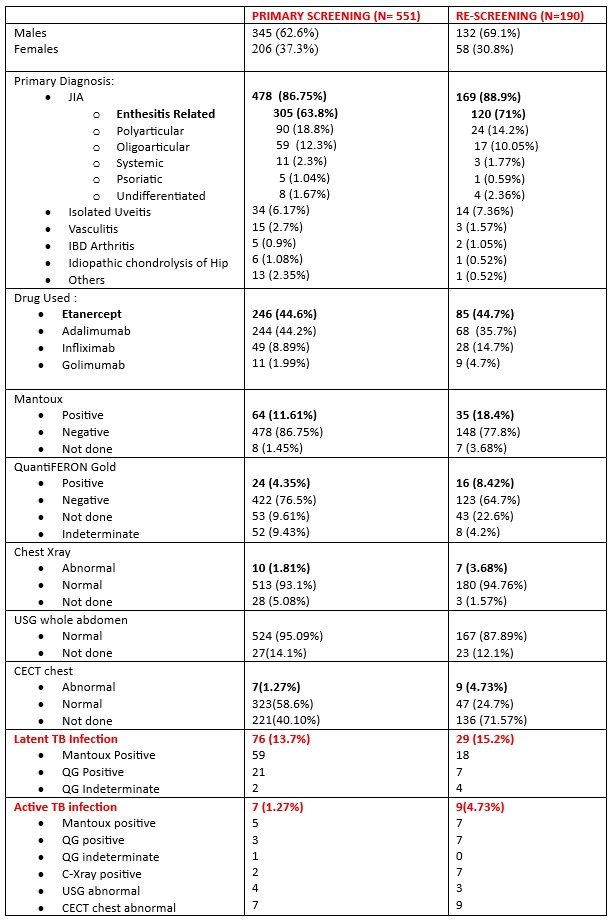
Conversion Rates for Each TNF Inhibitor on Rescreening 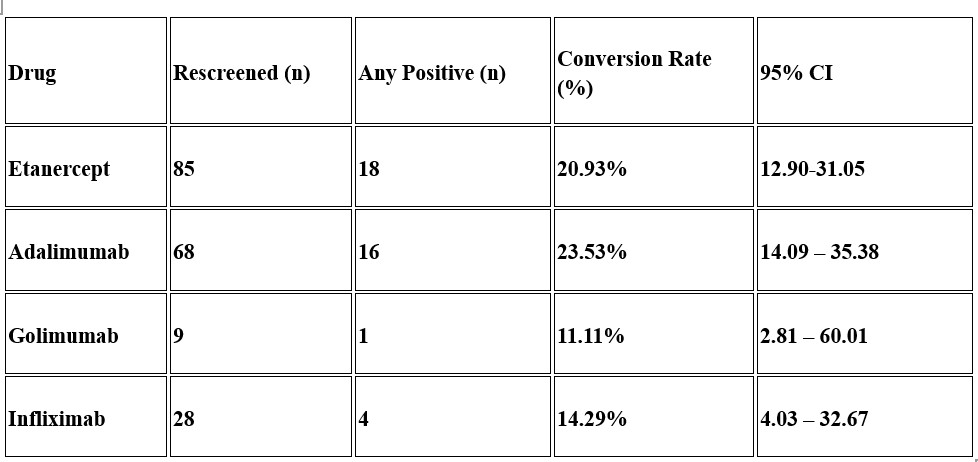 Adalimumab and Etanercept showed a higher conversion rate. Golimumab and Infliximab had lesser conversion rates. These conversion rates included both LTBI and Active TB cases.
Adalimumab and Etanercept showed a higher conversion rate. Golimumab and Infliximab had lesser conversion rates. These conversion rates included both LTBI and Active TB cases.
Concordance of Mantoux and QuantiFERON Gold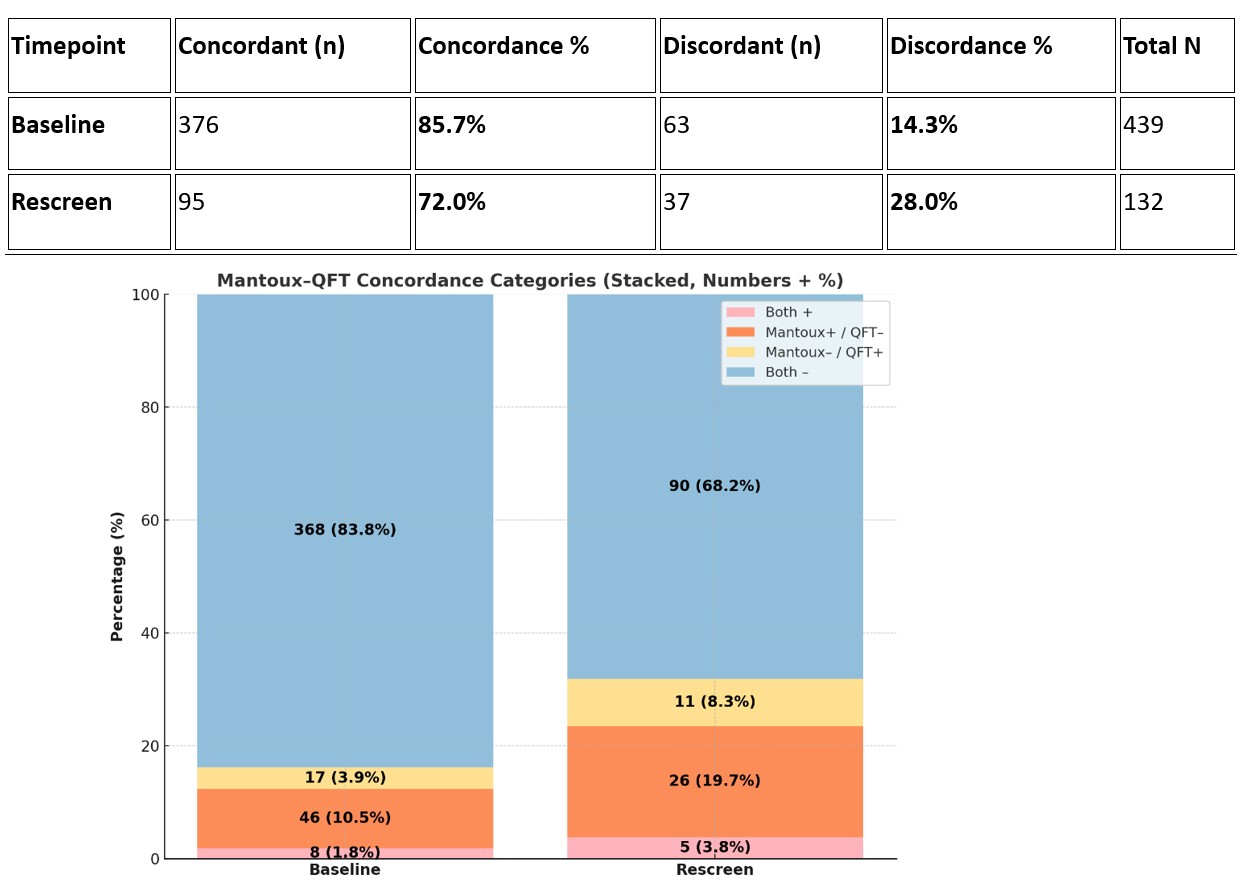 Mantoux–QuantiFERON results could be ascertained in 439/551 children at baseline and 132/190 at follow-up. Of these, discordance occurred in 63 (14.3%) vs 37 (28%).
Mantoux–QuantiFERON results could be ascertained in 439/551 children at baseline and 132/190 at follow-up. Of these, discordance occurred in 63 (14.3%) vs 37 (28%).
To cite this abstract in AMA style:
Rajpal N, Somashekar M, Agarwal m, Sawhney s. Burden of Latent and Active TB in Pediatric TNF-Inhibitor Therapy: A Decade of Real-World Data from a Tertiary Care Centre [abstract]. Arthritis Rheumatol. 2026; 78 (suppl 3). https://acrabstracts.org/abstract/burden-of-latent-and-active-tb-in-pediatric-tnf-inhibitor-therapy-a-decade-of-real-world-data-from-a-tertiary-care-centre/. Accessed .« Back to 2026 Pediatric Rheumatology Symposium
ACR Meeting Abstracts - https://acrabstracts.org/abstract/burden-of-latent-and-active-tb-in-pediatric-tnf-inhibitor-therapy-a-decade-of-real-world-data-from-a-tertiary-care-centre/
