Session Information
Session Time: 6:00PM-7:00PM
Background/Purpose: Chronic Nonbacterial Osteomyelitis (CNO) is a rare inflammatory bone disease in children that leads to significant functional impairment, growth defects, and adverse physical and psychosocial outcomes. Understanding the effectiveness of various treatment strategies is crucial for improving patient care. Previous MRI-based studies in CNO have not examined specific lesion characteristics, focusing mainly on changes in lesion count. Only one previous study evaluated the impact of aggressive therapy on disease activity and the risk of skeletal damage using MRI findings (1). Our study aims to retrospectively assess changes in disease activity and skeletal damage in pediatric CNO patients and evaluate the effects of different treatment regimens on disease progression and patient outcomes.
Methods: This retrospective study analyzed data from pediatric patients aged 2-21 years diagnosed with CNO between 2016 and 2025 at the Wash U/St. Louis Children’s Hospital (SLCH) pediatric rheumatology clinic. Patient identification was conducted through the hospital’s EPIC system. Key data points included demographics, clinical presentation, radiographic findings, and treatment regimens. MRI scans, conducted in the past, were analyzed retrospectively to assess disease activity and skeletal damage before and after treatment. Complete clinical response is defined as the resolution of pain, a normal physical exam, and a normal ESR, while partial clinical response is defined as an improvement in all these areas, but not a complete resolution.
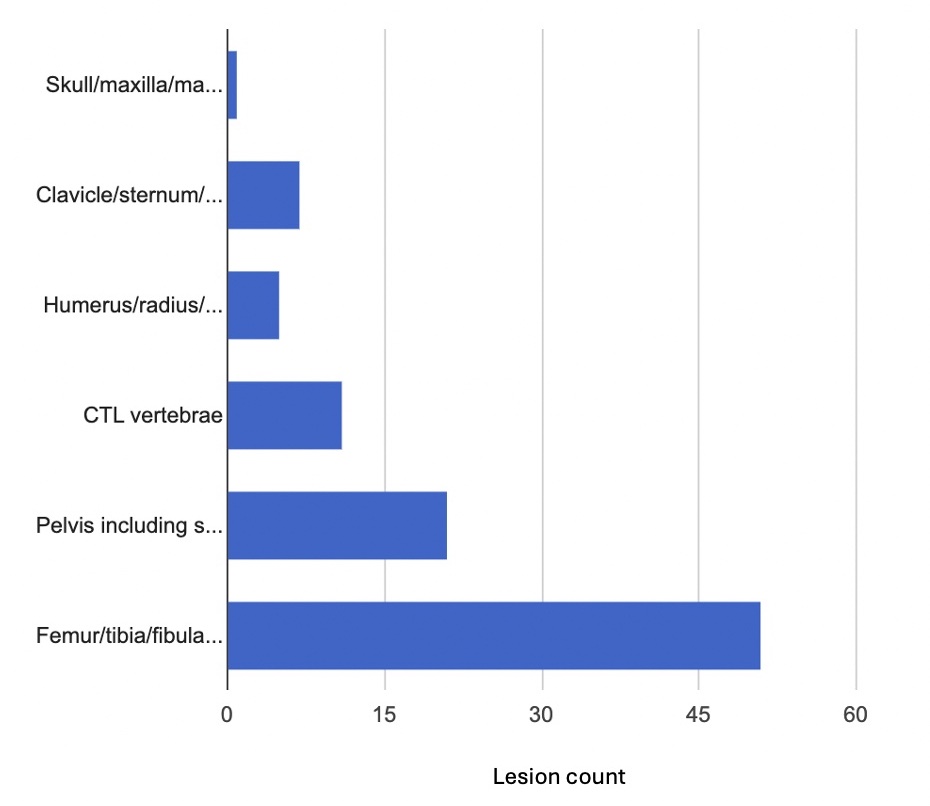
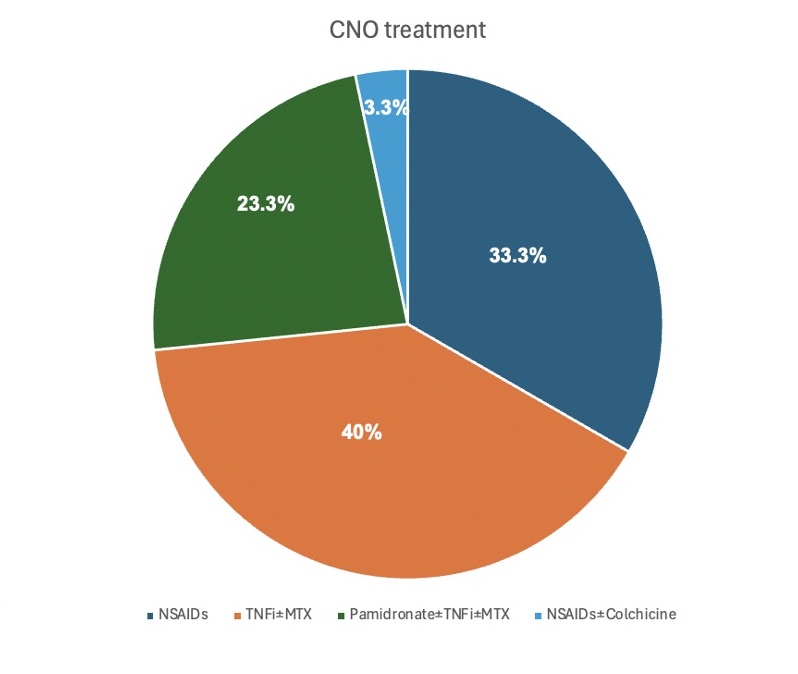
Results: Of the 75 identified patients, 30 met the inclusion criteria. The remaining 45 were excluded due to the absence of pretreatment and/or posttreatment MRI data. Within the study group, 54.8% were female, 45.2% were male, and 93% were white. At baseline, lesions were distributed as follows: 51 in femur, tibia, and fibula; 21 in pelvis (including sacrum and coccyx); 11 in cervical, thoracic, and lumbar vertebrae; 5 in humerus, radius, ulna, and hand; 7 in clavicle, sternum, scapula, and ribs; and 1 in mandible. (Fig 1) Twenty patients needed advanced treatments: 12 received TNF inhibitor (TNFi) with/without methotrexate, 7 received pamidronate with/without TNFi/methotrexate, and 1 received colchicine. (Fig 2) In the NSAIDs group (10 patients), 1 had a complete clinical response, 4 had partial clinical responses, and 5 had no improvement. In the advanced treatment group, all responded: 7 had complete clinical responses, and 13 had partial clinical responses. (Fig 3)
Conclusion: This study highlights the significant burden of CNO on pediatric patients and the necessity for diverse treatment regimens to manage the disease effectively. The data suggest advanced treatments, including TNFi, methotrexate, and pamidronate, are used in most patients. Further analysis of MRI scans will provide additional insights into the effectiveness of these treatment approaches, helping to optimize therapeutic strategies and improve patient outcomes.
References:
1- Zhao Y, Chauvin NA, Jaramillo D, Burnham JM. Aggressive Therapy Reduces Disease Activity without Skeletal Damage Progression in Chronic Nonbacterial Osteomyelitis. J Rheumatol. 2015;42(7):1245-1251.Disclosures: None
Figure 3: Clinical responses to the different treatment regimens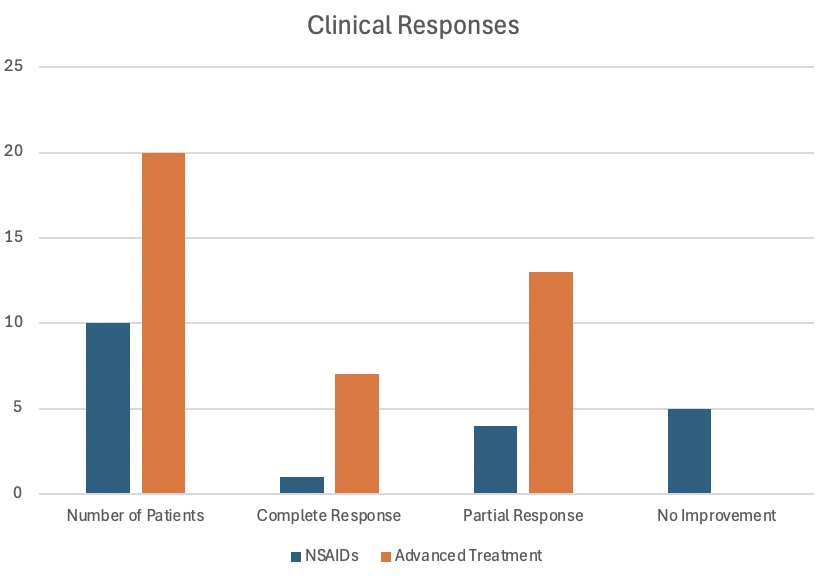
To cite this abstract in AMA style:
Civilibal Tang N, Marin Concha J, Baszis K, Zhao Y. Assessment of Disease Activity and Skeletal Damage in Pediatric Chronic Nonbacterial Osteomyelitis and the Effects of Different Treatment Regimens [abstract]. Arthritis Rheumatol. 2026; 78 (suppl 3). https://acrabstracts.org/abstract/assessment-of-disease-activity-and-skeletal-damage-in-pediatric-chronic-nonbacterial-osteomyelitis-and-the-effects-of-different-treatment-regimens/. Accessed .« Back to 2026 Pediatric Rheumatology Symposium
ACR Meeting Abstracts - https://acrabstracts.org/abstract/assessment-of-disease-activity-and-skeletal-damage-in-pediatric-chronic-nonbacterial-osteomyelitis-and-the-effects-of-different-treatment-regimens/