Session Information
Date: Saturday, March 21, 2026
Title: Plenary Abstract Session III
Session Time: 11:30AM-11:45AM
Background/Purpose: Adalimumab is the most widely used biologic disease-modifying antirheumatic drug (DMARD) for JIA. Many patients and families are interested in stopping medication for well-controlled JIA, but the optimal approach to stopping adalimumab is unclear.
Methods: We conducted a retrospective cohort study using data from the CARRA Registry (2016-2022). Eligible participants were taking adalimumab and had inactive non-systemic JIA (joint count=0, physician global assessment (PhGA)≤1, stable DMARD dose, no glucocorticoids) for 2 visits ≥5 months apart. Two deprescribing strategies (abrupt stop or tapering) were compared with adalimumab continuation. Participants were followed across multiple treatment strategies (e.g., continuing, tapering, stopping). Outcome #1 was flare (defined by higher adalimumab dose/frequency, restarted adalimumab after stopping, new DMARD or glucocorticoid, joint count ≥1, PhGA >1, or new uveitis activity). Among those who flared, outcome #2 was recapture of inactive disease. Causal associations between treatment strategy and outcomes were estimated using marginal structural modeling with weighted Cox proportional hazards regression and time-varying treatment. Stabilized inverse probability of treatment weights (IPTWs) were constructed from treatment models stratified by prior treatment state, adjusted for clinical, disease-related, and treatment covariates (including concomitant methotrexate use) and expressed as adjusted hazard ratios (aHRs) with 95% confidence intervals (CIs). Missing covariates were handled using multiple imputation by chained equations.
Results: A total of 860 eligible participants were included (mean age 11.8, 67% female, 63% with oligoarthritis or RF-negative polyarthritis) (Table 1). Of these, 50 (5.8%) abruptly stopped after continuation, while 125 (14.5%) tapered, and 213 (24.8%) stopped after taper. Tapers took place over a median of 201 days (interquartile range 121, 301). Overall, 364 flares were documented, occurring at a median of 406 days after the start of follow-up. Compared with continuation, abruptly stopping adalimumab was associated with higher rates of subsequent flare (aHR 2.48, 95% CI 1.87, 3.28), but tapering was not (aHR 1.27, 95% CI 0.80, 2.03) (Table 2, Figure). However, rates of flare were even higher after stopping post-taper (aHR 4.96, 95% CI 3.40, 7.24) (Table 2, Figure). Among those who flared, adjusted rates of recapture were numerically higher during, but not after, tapering (Table 2). However, estimates were imprecise, and results were not significantly different across strategies.
Conclusion: In children and youth with well-controlled JIA, tapering adalimumab led to similar rates of flare compared to continuation. However, those who stopped adalimumab after taper flared sooner than those who stopped without taper, suggesting a trade-off between better short-term outcomes while tapering and worse long-term outcomes after tapering. These results and their underlying mechanism (e.g., anti-drug antibody formation) warrant further investigation in other populations.
Table 1: Baseline characteristics of children with well-controlled JIA on adalimumab in the CARRA Registry.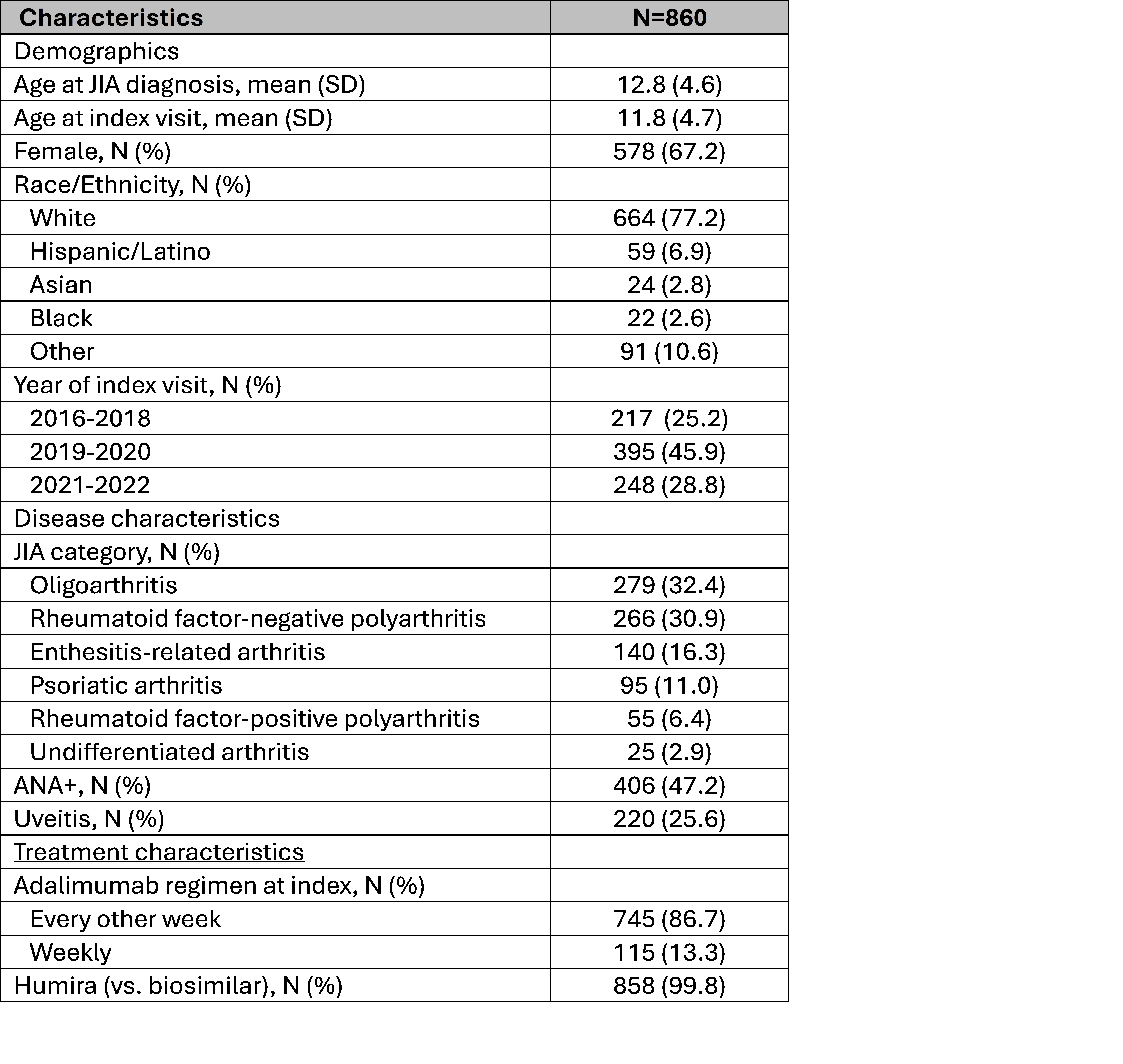 ANA, antinuclear antibody; bDMARD, biologic disease-modifying anti-rheumatic drug; cDMARD, conventional disease-modifying anti-rheumatic drug; SD, standard deviation
ANA, antinuclear antibody; bDMARD, biologic disease-modifying anti-rheumatic drug; cDMARD, conventional disease-modifying anti-rheumatic drug; SD, standard deviation
Table 2. Causal associations between deprescribing strategy and outcomes of flare and recapture of inactive disease.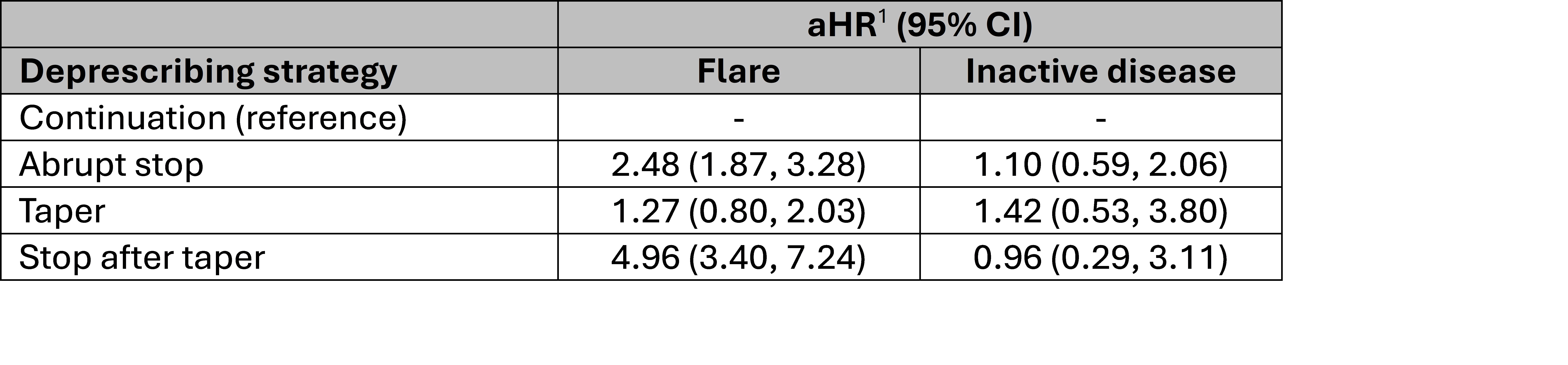 1, Estimates from marginal structural modeling with weighted Cox proportional hazards regression and time-varying treatment, adjusted for age, sex, race, ethnicity, standardized height and weight, JIA type, joint involvement (oligo vs. poly), ANA status, history of uveitis, Childhood Health Assessment Questionnaire (CHAQ), patient/caregiver global score, and concomitant conventional disease-modifying anti-rheumatic drug (DMARD) use. Additional covariates for inactive disease models were physician global score, patient/caregiver global score, and active joint count at flare, and time from deprescribing (or continuation) to flare.
1, Estimates from marginal structural modeling with weighted Cox proportional hazards regression and time-varying treatment, adjusted for age, sex, race, ethnicity, standardized height and weight, JIA type, joint involvement (oligo vs. poly), ANA status, history of uveitis, Childhood Health Assessment Questionnaire (CHAQ), patient/caregiver global score, and concomitant conventional disease-modifying anti-rheumatic drug (DMARD) use. Additional covariates for inactive disease models were physician global score, patient/caregiver global score, and active joint count at flare, and time from deprescribing (or continuation) to flare.
Figure. Model-adjusted survival without flare based on hypothetical treatment strategies.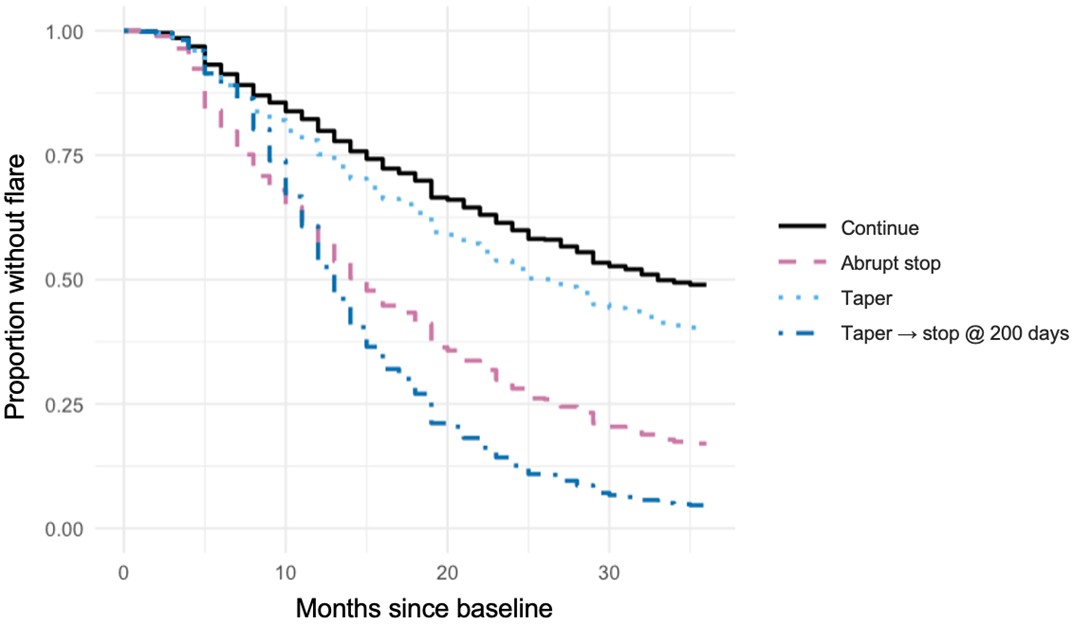 Model-adjusted Kaplan-Meier curves for survival without flare for hypothetical treatment strategies (continue, abrupt stop, taper, taper and then stop after 200 days, the median duration of observed tapers), estimated using marginal structural modeling with weighted Cox proportional hazards regression and time-varying treatment, adjusted for age, sex, race, ethnicity, standardized height and weight, JIA type, joint involvement (oligo vs. poly), ANA status, history of uveitis Childhood Health Assessment Questionnaire (CHAQ), patient/caregiver global score, and concomitant conventional disease-modifying anti-rheumatic drug (DMARD) use.
Model-adjusted Kaplan-Meier curves for survival without flare for hypothetical treatment strategies (continue, abrupt stop, taper, taper and then stop after 200 days, the median duration of observed tapers), estimated using marginal structural modeling with weighted Cox proportional hazards regression and time-varying treatment, adjusted for age, sex, race, ethnicity, standardized height and weight, JIA type, joint involvement (oligo vs. poly), ANA status, history of uveitis Childhood Health Assessment Questionnaire (CHAQ), patient/caregiver global score, and concomitant conventional disease-modifying anti-rheumatic drug (DMARD) use.
To cite this abstract in AMA style:
Horton D, Li Y, Nyandege A, Liu N, Luo L, Beukelman T, Feldman B, Mannion M, Sutton A, twilt M, Verstegen R, Hu L. Adalimumab Tapering is Associated with Short-term, but not Long-term, Advantages for Well-controlled Juvenile Idiopathic Arthritis: A Retrospective Cohort Study using the CARRA Registry [abstract]. Arthritis Rheumatol. 2026; 78 (suppl 3). https://acrabstracts.org/abstract/adalimumab-tapering-is-associated-with-short-term-but-not-long-term-advantages-for-well-controlled-juvenile-idiopathic-arthritis-a-retrospective-cohort-study-using-the-carra-registry/. Accessed .« Back to 2026 Pediatric Rheumatology Symposium
ACR Meeting Abstracts - https://acrabstracts.org/abstract/adalimumab-tapering-is-associated-with-short-term-but-not-long-term-advantages-for-well-controlled-juvenile-idiopathic-arthritis-a-retrospective-cohort-study-using-the-carra-registry/
