Session Information
Session Time: 6:00PM-7:00PM
Background/Purpose: Pathogenic variants in RELA, encoding the p65 (RelA) protein, a key member of the canonical NFkB pathway, have been linked to chronic mucocutaneous inflammatory features, including inflammatory bowel disease (IBD) and Behçet’s disease (BD). We analyzed the clinical phenotypes of a large, international, multicenter cohort of patients with RelA haploinsufficiency to describe the phenotypic variations and perform genotype-phenotype correlations for this disease.
Methods: To identify patients, we contacted physicians through an international email list to determine if they were caring for symptomatic patients with an identified RELA variant. Collaborating practitioners provided deidentified clinical, laboratory, imaging, treatment, and outcomes data. Family members identified with a familial RELA variant with or without symptoms were also included.
Results: Forty patients were enrolled in the study, ranging in age from 12 months to 62 years old at the time of enrollment. The mean age at onset of symptoms was 9.9 years, while the mean age at diagnosis was 16.5 years. The most common manifestations of disease (Table 1) were oral ulcers (60%); GI manifestations including IBD and eosinophilic esophagitis (58%); arthritis or arthralgia (48%); skin problems including psoriasis, erythema nodosum, recurrent ulcers or pustules, atopic dermatitis, urticaria, or malar rash (43%); genital ulcers (36%); and ophthalmic problems including uveitis, episcleritis, or conjunctivitis (20%). Of note, five patients had malignancy, including two with lymphoma (33-year-old and 28-year-old males; neither had preceding exposure to biologic medications) and two with cervical cancer.
Immunologic evaluation (Table 1) revealed that 43% of the cohort had a positive ANA, though only 15% had any other positive autoantibody. One patient presented with lupus manifestations including malar rash, arthritis, lymphopenia, positive ANA (1:1280, homogenous), positive dsDNA, positive Coombs, positive ribosomal P antibody, and positive Smith antibody.
Treatments included (Table 2) glucocorticoids, nonbiologic DMARDs such as colchicine, TNF inhibitors, JAK inhibitors, apremilast, interleukin inhibitors, integrin inhibitors, and B-cell depleting agents. Four patients (10%) required no treatment.
Conclusion: We report one of the largest global cohorts of RelA haploinsufficiency to date. All patients with a clinical phenotype demonstrated one or more inflammatory features, with a range of phenotypes beyond the common mucocutaneous manifestations, with possible increased risk for malignancy. There was evidence of incomplete penetrance and variable expressivity. Studies on the functional impact of the specific RELA variants, genotype-phenotype correlations, and immunophenotyping are currently underway. We hope to continue enrolling patients to clarify whether these characteristics are enriched compared to background rates, compare co-incidence of clinical features to known entities such as BD, and to characterize the evolution of disease over the lifespan. Thus, this cohort represents a rich dataset to explore the epidemiology and clinical phenotype of a very rare inborn error of immunity.
Table 1: Demographics of the RelA Haploinsufficiency Cohort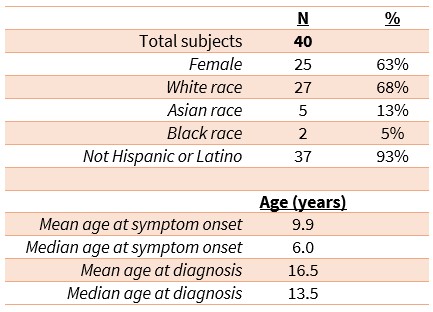
Table 2: Clinical Characteristics and Autoimmune Workup of the RelA Haploinsufficiency Cohort
Treatments Tried in the RelA Haploinsufficiency Cohort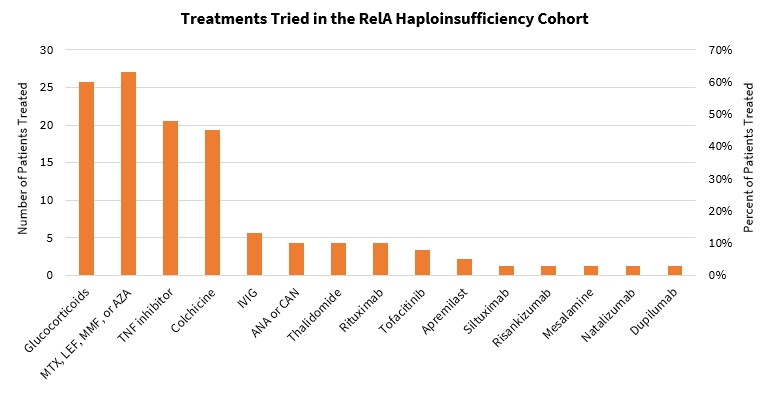 MTX = methotrexate, LEF = leflunomide, MMF = mycophenolate, AZA = azathioprine, ANA = anakinra, CAN = canakinumab
MTX = methotrexate, LEF = leflunomide, MMF = mycophenolate, AZA = azathioprine, ANA = anakinra, CAN = canakinumab
To cite this abstract in AMA style:
Murray E, Akoghlanian S, Dileepan G, Guo L, Allenspach E, Alsulami S, Alsuweiti M, Ardalan K, Broderick L, Cebecauerova D, Chan R, Chapdelaine H, D'Astous-Gauthier K, Dedeoglu F, Efron Srour A, Igel L, Lawrence M, Lee P, Maltz R, Naesens L, Ombrello a, Padem N, Paquin M, Peng X, Rosenwasser N, Saca L, Sánchez-Manubens J, Taxter A, Towers V, Wonnaparhown A, Zheng S, Zimmerman O, Abraham R, Sivaraman V. More Than Meets the Eye: Broadening the Clinical Spectrum of RelA Haploinsufficiency [abstract]. Arthritis Rheumatol. 2026; 78 (suppl 3). https://acrabstracts.org/abstract/more-than-meets-the-eye-broadening-the-clinical-spectrum-of-rela-haploinsufficiency/. Accessed .« Back to 2026 Pediatric Rheumatology Symposium
ACR Meeting Abstracts - https://acrabstracts.org/abstract/more-than-meets-the-eye-broadening-the-clinical-spectrum-of-rela-haploinsufficiency/
