Session Information
Date: Friday, March 20, 2026
Title: Abstracts: Technology
Session Time: 4:55PM-5:00PM
Background/Purpose: Strategies to facilitate blood collection outside of clinical spaces may enhance the participation and satisfaction of participants in clinical and translational research studies. We examined the feasibility, acceptability, and reliability of self-/caregiver-collected capillary blood samples via a button device (Tasso) in individuals with JIA on adalimumab.
Methods: Pilot of Adalimumab Withdrawal (PAW) is a prospective observational study conducted within the CARRA Registry. Selected registry participants with JIA on adalimumab consented to participate in the PAW study, where they provided venous and capillary blood samples at a baseline visit in clinic (PAW baseline). A second capillary blood sample was collected 4-8 weeks later at participants’ homes. Participants or their caregivers completed questionnaires about their acceptability of the Tasso devices after each use. To examine the feasibility of lower-cost shipping, capillary blood samples were sent to the CARRA Biobank from sites and homes via regular postal service. Serum volumes obtained from devices and viability were described, and questionnaire responses were analyzed using median and interquartile range (IQR).
Results: Between May 2023 and May 2025, 30 individuals enrolled in PAW (Table 1) and underwent 54 capillary blood sample collections; 6 participants missed follow-up sampling. The devices collected a median of 200 µL serum/sample (IQR 90-300 µL, min-max 0-430 µL). 41 of 54 sample collection attempts yielded adequate serum samples (75.9%). Device failure (i.e., inability to obtain any blood) occurred in 4 instances (7.4%), and one sample went missing in the postal service (1.9%). In addition, serum separations were unsuccessful in 8 samples (14.8%), which were associated with longer sample transit times (median 8.5 vs 6 days, p=0.01) and lower minimum temperatures at the day of arrival at the CARRA Biobank (Cincinnati, OH; median -8.9 vs 6.1 °C, p< 0.001; Figure 1). Overall, most participants and families reported minimal pain and high acceptability of Tasso devices at baseline and during the at-home collection (Table 2). However, higher pain scores were reported by children < 11 years old compared to older participants (Kruskal-Wallis Test, p< 0.001).
Conclusion: The feasibility and acceptability of Tasso devices appear promising. However, several limitations were identified in this pilot study, including device failures in nearly 1 in 10 attempts, negative effects on sample separation caused by colder weather and prolonged transit times, and higher levels of pain among younger children. More data are needed to understand whether laboratory measurements within serum are comparable between capillary (Tasso) and venous blood samples.
Table 1. Baseline characteristics of enrolled PAW participants.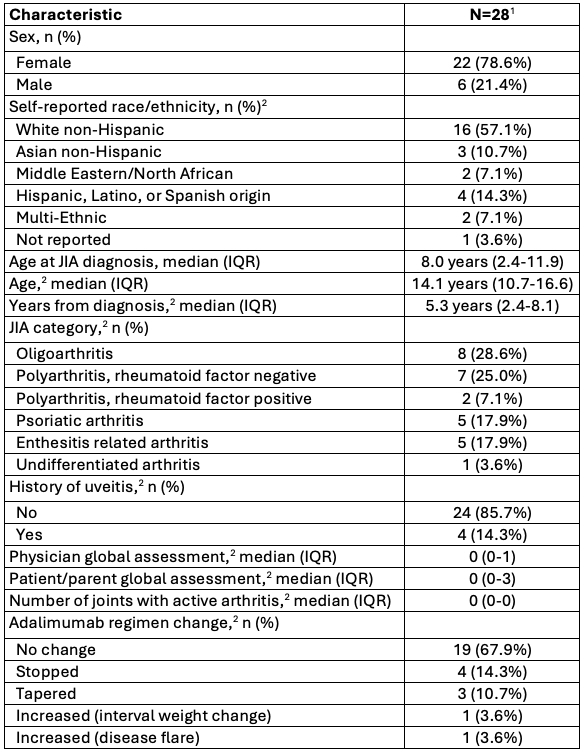 IQR, interquartile range. 1, Two participants without data collected from baseline visit. 2, At PAW enrollment.
IQR, interquartile range. 1, Two participants without data collected from baseline visit. 2, At PAW enrollment.
Table 2. PAW acceptability questionnaire results by visit and age group.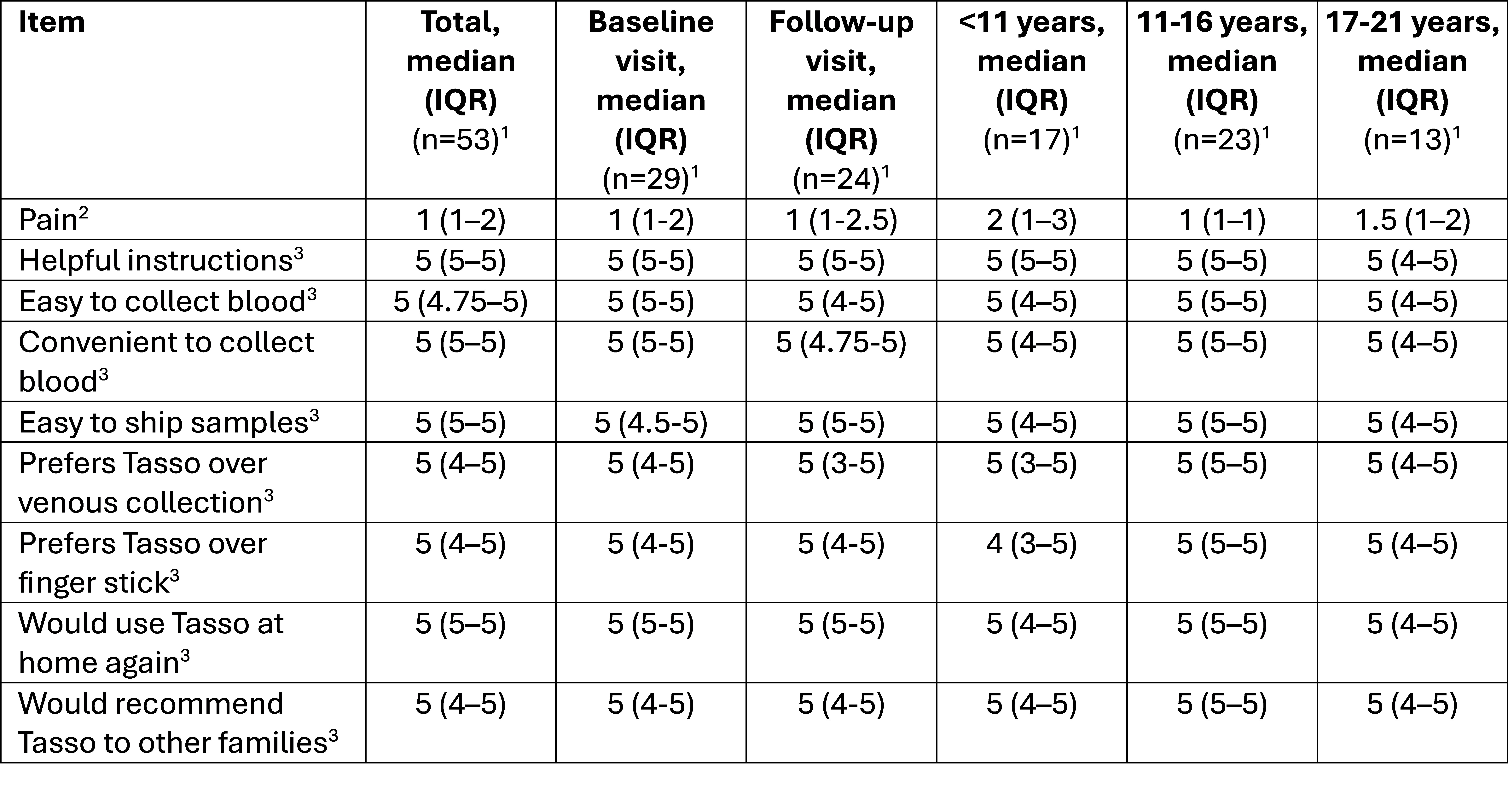 1, Number of assessments. 2, Range from (1) no pain to (5) severe pain. 3, Range from (1) completely disagree to (5) completely agree.
1, Number of assessments. 2, Range from (1) no pain to (5) severe pain. 3, Range from (1) completely disagree to (5) completely agree.
Figure 1: Effect of transit time and outside temperature on successful serum separation.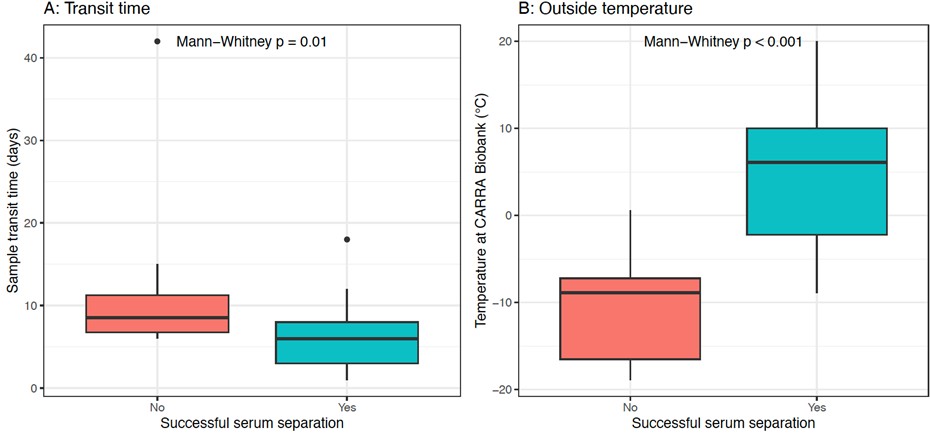 Boxplots show the effect of (A) sample transit time (days) and (B) outside temperature (°C) on successful serum separation. Red: No successful separation; Green: Successful separation. Mann–Whitney p-values indicate statistical significance.
Boxplots show the effect of (A) sample transit time (days) and (B) outside temperature (°C) on successful serum separation. Red: No successful separation; Green: Successful separation. Mann–Whitney p-values indicate statistical significance.
To cite this abstract in AMA style:
Verstegen R, Iizuka A, Godiwala C, Iozzio M, Kohlheim M, Park K, Russell A, Sudman m, Sutton A, Balay-Dustrude E, Cooper j, Drew J, Sagcal-Gironella A, Goh Y, Halyabar O, McBrearty K, Moorthy L, Nowakowski A, Oakes R, oberle E, Riordan M, twilt M, Varghese C, Horton D. Pilot of Adalimumab Withdrawal (PAW): Preliminary Analysis of Feasibility and Acceptability of Self-Collected Capillary Blood Samples [abstract]. Arthritis Rheumatol. 2026; 78 (suppl 3). https://acrabstracts.org/abstract/pilot-of-adalimumab-withdrawal-paw-preliminary-analysis-of-feasibility-and-acceptability-of-self-collected-capillary-blood-samples/. Accessed .« Back to 2026 Pediatric Rheumatology Symposium
ACR Meeting Abstracts - https://acrabstracts.org/abstract/pilot-of-adalimumab-withdrawal-paw-preliminary-analysis-of-feasibility-and-acceptability-of-self-collected-capillary-blood-samples/
