Session Information
Session Time: 5:00PM-6:00PM
Background/Purpose:
Emapalumab is a human interferon gamma antibody that was FDA approved to treat MAS secondary to sJIA in June 2025. It was previously approved for primary hemophagocytic lymphohistiocytosis treatment, targeting IFN-driven hyperinflammation. Several studies have investigated the safety of emapalumab and its efficacy for sJIA, including long-term follow up to 12 months.
This study summarizes our institution’s experience of the efficacy and safety of emapalumab treatment for sJIA with recurrent MAS, for disease flares and remission maintenance.
Methods:
With institutional review board approval, a retrospective chart review of sJIA patients treated with emapalumab for recurrent MAS was performed from January 2021 through June 30, 2024, based on IRB study period. Patient demographics, clinical features, treatments and outcomes were analyzed using standard descriptive statistics.
Results:
Four patients were included with mean age of 2.9 years at diagnosis and 5.2 years at emapalumab initiation (Table 1). At time of initiation, all were receiving anti IL-1 agents, with most receiving concomitant JAK inhibitors, and 3 had developed sJIA-associated lung disease. All patients started with twice weekly emapalumab dosing. Duration of emapalumab treatment ranged from 3.8 to 42 months. By the end of the study, 2 patients had weaned to physiologic steroid doses, while the other 2 patients were completely off steroids 6-8 months after emapalumab initiation.
Table 2 summarizes adverse effects and the presence or progression of sJIA-associated lung disease during treatment. Of the 3 patients who had new or worsening lung imaging findings during treatment, only Patient 2’s findings were confirmed to be related to sJIA, and Patient 2 had improvement in respiratory symptoms during the study period. Patient 3 developed asymptomatic pneumocystis jrovecii (PJP) during treatment while off prophylaxis due to gastrointestinal side effects. During emapalumab treatment, no patient developed infections with Epstein-Barr Virus (EBV), cytomegalovirus (CMV), or herpes virus (HSV), significant cytopenias, or transaminitis.
All patients experienced reductions in IL-18, CXCL-9, and ferritin by study end, yet IL-18 levels did not fully normalize (Table 3).
Conclusion:
This review demonstrates the efficacy and safety of emapalumab for induction and maintenance of remission in sJIA with recurrent MAS. All patients showed improvement in systemic features and key inflammatory markers. Despite overall clinical improvement, one patient developed a slight increase in lung opacities on CT after 1 month of emapalumab. The most significant adverse event was an asymptomatic PJP infection. All patients received prophylactic antimicrobials and were closely monitored throughout treatment. Notably, Patient 1 has remained on emapalumab for over 3.5 years. Attempts to taper therapy either by reducing the dose or extending the dosing interval led to recurrence of MAS features, though full-blown MAS was prevented with dose adjustments. This study highlights a potential role for empalumab beyond its approved use suggesting benefit in some patients with refractory sJIA and that it may serve as a bridge for bone marrow transplant.
Table 1: Patient Characteristics and Clinical Course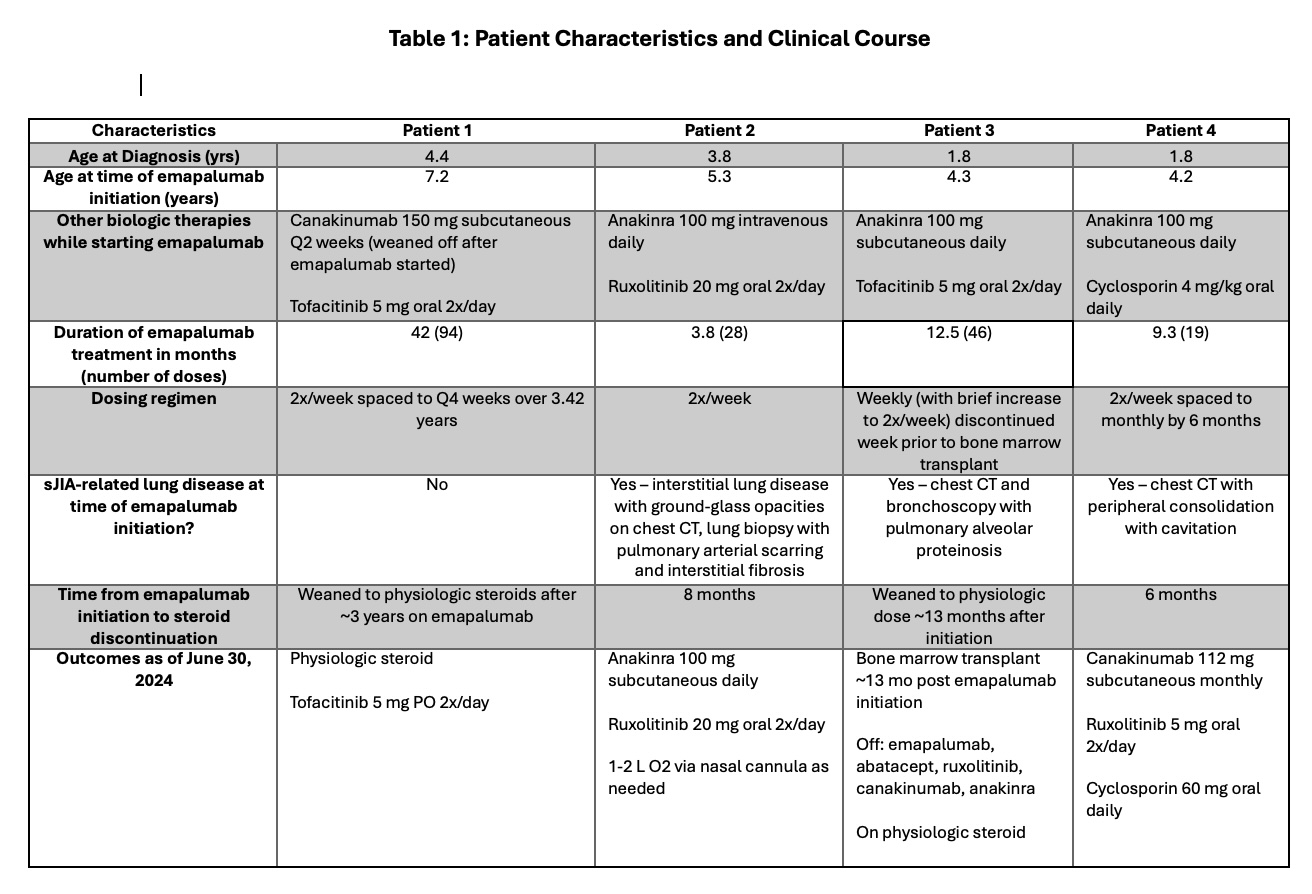
Table 2: Adverse Effects and Safety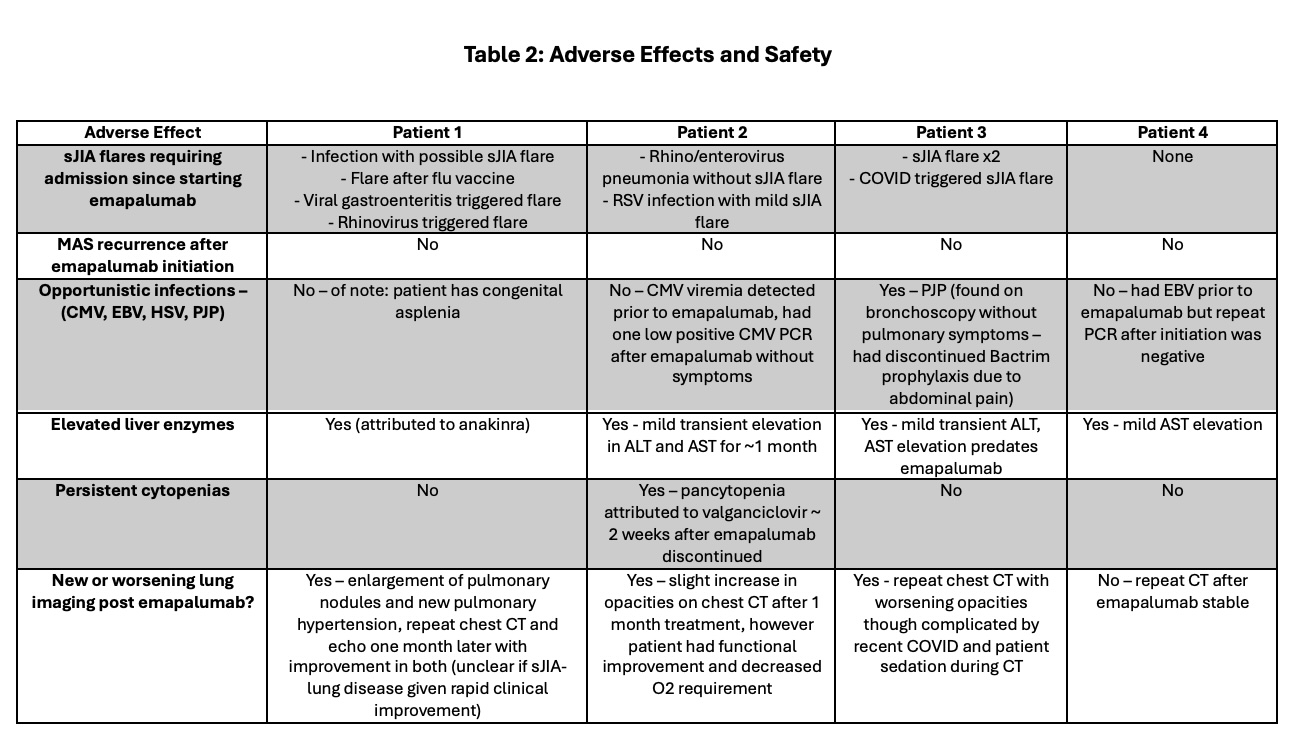
Table 3: MAS Inflammatory Markers Following Emapalumab Initiation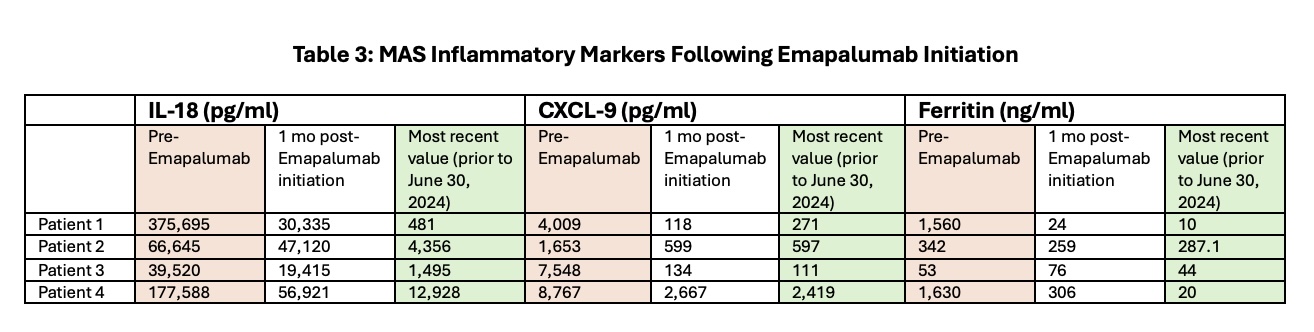
To cite this abstract in AMA style:
Butts M, DeGuzman M, McClain K, Butler E, Ramirez A. Targeting Macrophage Activation Syndrome (MAS) in Systemic Juvenile Idiopathic Arthritis (sJIA): Real-World Emapalumab Experience Spanning 3 Years [abstract]. Arthritis Rheumatol. 2026; 78 (suppl 3). https://acrabstracts.org/abstract/targeting-macrophage-activation-syndrome-mas-in-systemic-juvenile-idiopathic-arthritis-sjia-real-world-emapalumab-experience-spanning-3-years/. Accessed .« Back to 2026 Pediatric Rheumatology Symposium
ACR Meeting Abstracts - https://acrabstracts.org/abstract/targeting-macrophage-activation-syndrome-mas-in-systemic-juvenile-idiopathic-arthritis-sjia-real-world-emapalumab-experience-spanning-3-years/
