Session Information
Session Time: 5:00PM-6:00PM
Background/Purpose:
Infliximab is an effective treatment for Kawasaki Disease (KD) as part of intensified initial therapy in combination with intravenous immunoglobulin (IVIG) or as a second-line medication. However, second-dose infliximab (10 mg/kg) for refractory patients has not been well-described. This study characterizes patients receiving two infliximab doses for KD within a 3-week period and describes safety outcomes.
Methods: We conducted a retrospective case series of children with KD (American Heart Association criteria) hospitalized at Children’s Hospital of Colorado between 2017 and 2025 and treated with IVIG and two doses of infliximab (10mg/kg/dose), aspirin, +/- additional therapies. Descriptive statistics were used for cohort analysis. Charts were reviewed through 12-13 months after discharge.
Results:
Thirty-four patients met inclusion criteria, 20 (59%) with complete KD and 14 (41%) with incomplete KD. Median age was 2.25 years (interquartile ratio 0.9, 4.75), a majority were male (65%), and they primarily identified as Hispanic/Latino (44%) or non-Hispanic White (34%). See Table 1 for demographics/clinical features. Thirty-one of 34 patients received initial intensified therapy with both IVIG and infliximab within 24-hours of starting either therapy; 3 patients received IVIG alone [Figure 1]. The interval between 1st and 2nd-dose infliximab ranged from 1-19.6 days, with a median of 2.4 (1.9, 3.0) days. Four patients received 2nd-dose IVIG before or concurrently with 2nd-dose infliximab. After 2nd-dose infliximab, 9 patients required additional treatments: anakinra(1), steroids (2), steroids + anakinra (2), and 2nd-dose IVIG (4). Pretreatment echo showed 4 (12%) patients had normal left anterior descending and right coronary artery Z-scores; 7 (20.5%), 17 (50%), and 6 (17.5%) had Z-scores between 2.00-2.49, 2.50-4.99, and 5.00-9.99, respectively. None had giant aneurysms at baseline; 4 later did with 1 with persistence at last follow up. One patient never developed coronary artery abnormalities (CAA). Last available follow up echo (n=11, 9, and 14 at 11-13 months, 7-10 months, and < 7 months, respectively) showed no CAA in 22 (65%) [Figure 1]. Adverse events [Table 2] while hospitalized included aseptic meningitis (n=2) and infusion reaction (n=2), all attributed temporally to IVIG. Neutropenia developed in 13 (38%) with 2 (6%) severe (absolute neutrophil count < 500). Within 12-13 months of discharge, 11 patients experienced 16 infections including 4 episodes of acute otitis media. No patient had reactivation of tuberculosis, endemic fungal disease, or other serious infections.
Conclusion:
This series characterizes a cohort of patients with refractory KD treated with IVIG and two infliximab doses within a 3-week period. Second-dose infliximab was effective and well-tolerated, with no major safety concerns identified. Further studies are needed to evaluate the efficacy and safety of using a second dose of infliximab compared with other treatment modalities.
Table 1: Patient Demographics, Clinical Features, and Baseline Laboratory Features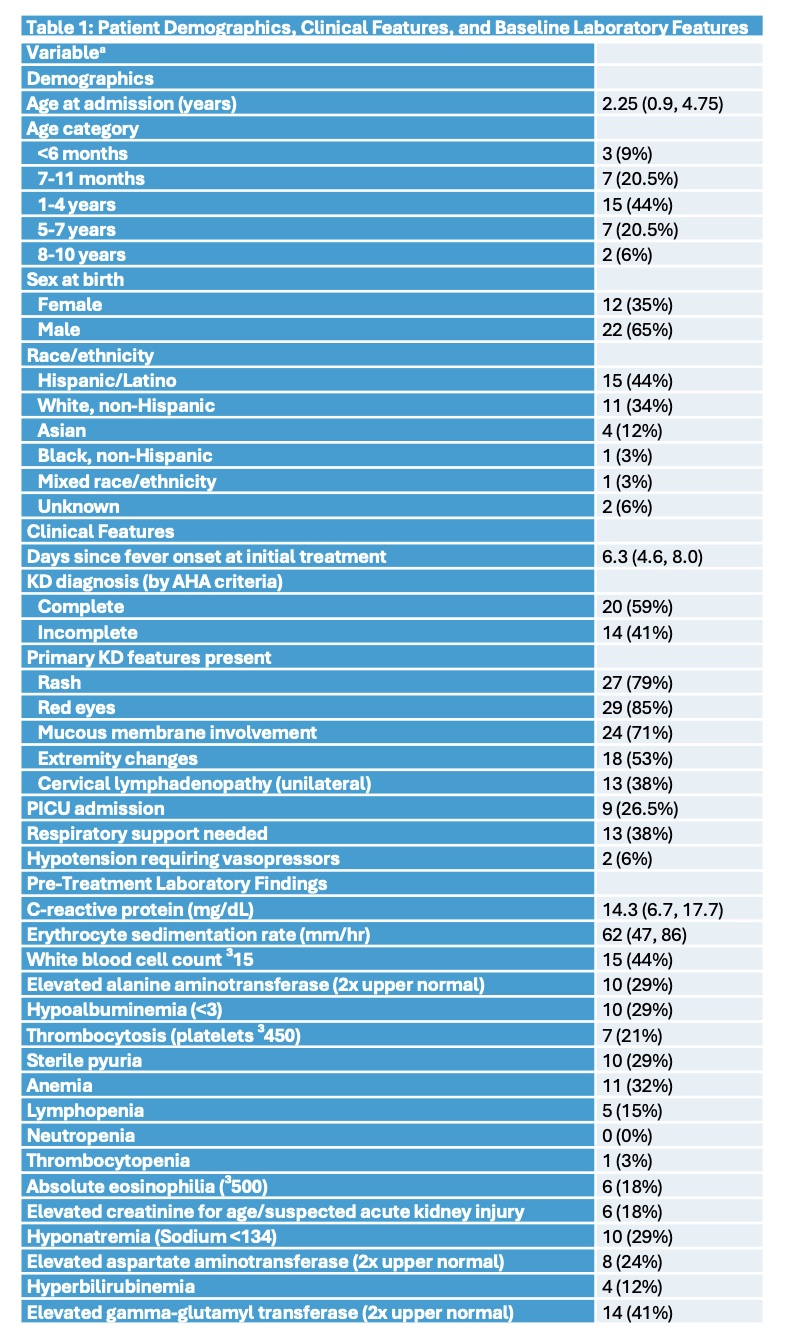 a Continuous variables presented as median (IQR); categorical variables presented as number (%). Total n=34 for each variable.
a Continuous variables presented as median (IQR); categorical variables presented as number (%). Total n=34 for each variable.
Table 2: Adverse Events Post-Treatment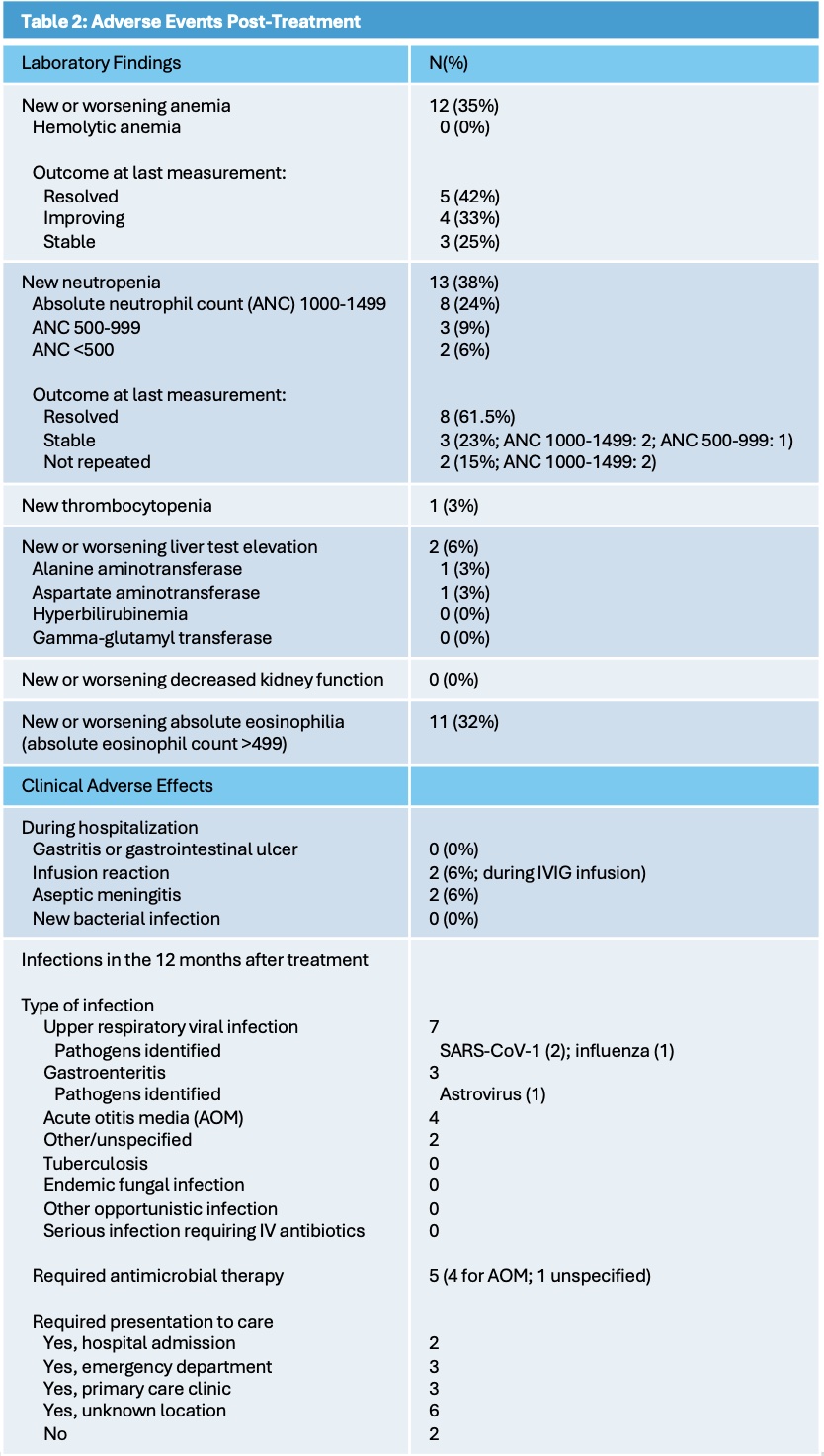
Figure 1: Timeline of Individual Patient Coronary Artery Abnormalities & Treatments Received 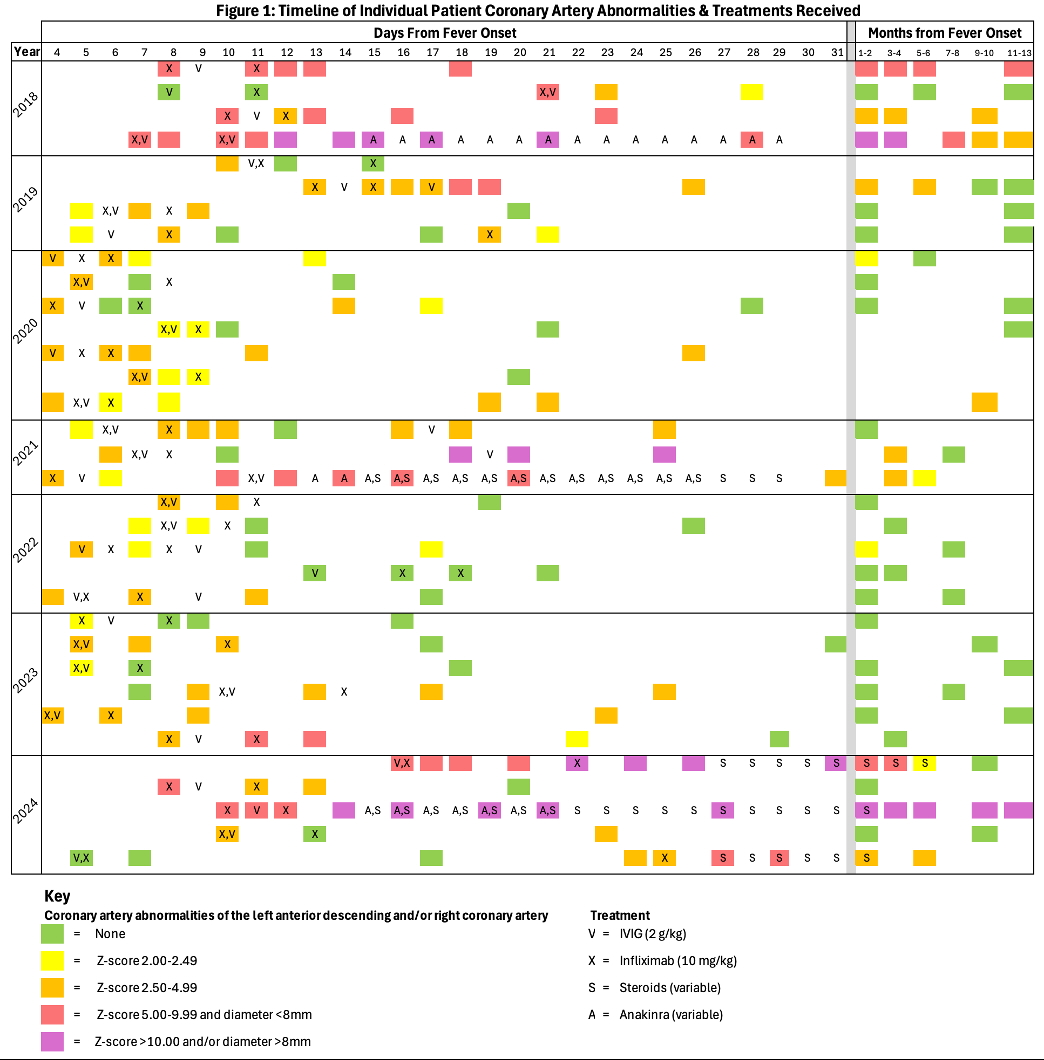
Methods: Each row represents an individual patient. Each shaded square designates that an echocardiogram (echo) was performed on that day of the course of illness from fever onset, with results of the echo color-coded by degree of coronary artery abnormalities as per the key. Treatments received by each patient are represented on this longitudinal timeline by letters as per the key to align with the day that they were received. When more than one therapy was given on a single calendar day, the letters are listed in the order they were administered. Of note, echos and treatments are entered on the calendar day (midnight to midnight) they were obtained; thus, the exact interval between individual echos and therapies cannot be assumed to be a full 24 hours when entered on consecutive days, for example. Summary: 31 of 34 patients received initial intensified therapy with both IVIG (V) and infliximab (X) within a 24 hours of starting either therapy; the remaining 3 patients received IVIG alone. The interval between first and second-dose infliximab ranged from 1-19.6 days, median 2.4 (IQR 1,9, 3.0). Four patients received second-dose IVIG before or concurrently with second-dose infliximab. After 2nd-dose infliximab, 9 patients required additional treatments: anakinra(1), steroids (2), steroids + anakinra (2), and 2nd-dose IVIG (4). Pretreatment echo showed 4 (12%) patients had normal left anterior descending and right coronary artery Z-scores; 7 (20.5%), 17 (50%), and 6 (17.5%) had Z-scores between 2.00-2.49, 2.50-4.99, and 5.00-9.99, respectively. None had giant aneurysms at baseline; 4 later did with 1 with persistence at last follow up. One patient never developed coronary artery abnormalities (CAA). Last available follow up echo (n=11, 9, and 14 at 11-13 months, 7-10 months, and < 7 months, respectively) showed Z-scores of < 2 (normal) in 26 (76%), 2-2.49 in 1 (3%), 2.5-4.99 in 5 (15%), 5-9.99 in 1 (3%), and 10 in 1 (3%).
To cite this abstract in AMA style:
Pavlenko M, Scheinberg K, Bartholomeo A, Lazaroff S, Levin C, Frappa K, Miller M, Vidas D, Anderson M, Dominguez S, Cole L. Clinical Characteristics and Safety Outcomes in Refractory Kawasaki Disease Treated with Second-Dose Infliximab [abstract]. Arthritis Rheumatol. 2026; 78 (suppl 3). https://acrabstracts.org/abstract/clinical-characteristics-and-safety-outcomes-in-refractory-kawasaki-disease-treated-with-second-dose-infliximab/. Accessed .« Back to 2026 Pediatric Rheumatology Symposium
ACR Meeting Abstracts - https://acrabstracts.org/abstract/clinical-characteristics-and-safety-outcomes-in-refractory-kawasaki-disease-treated-with-second-dose-infliximab/
