Session Information
Session Time: 5:00PM-6:00PM
Background/Purpose: Despite continuous improvement in care and the recent update of international recommendations, relevant discrepancies in the approach to Still’s disease (SD) still exists.The study was aimed to explore current global treatment strategies for SD focusing oninital management and refractory disease.
Methods: As part of the METAPHOR project, a PReS/PRINTO initiative to optimize therapy in SD and Macrophage Activation Syndrome (MAS), a global survey was developed. Topics were selected by 22 expert pediatric rheumatologists; the web-survey was completed between 3/12/24 and 14/2/25.
Results: .A total of 206 physicians (91% paediatric rheumatologists) from 56 countries completed the survey. Investigations performed at onset included infectious screening (80%), peripheral blood smears (85%), ferritin (100%), imaging ( 80%). Seventy % of clinician would perform additional screening tests if glucocorticoids had to be started. In newly diagnosed SD, 66% of clinicians would use GCs as 1st line (42% associated with an IL-1/IL-6 inhibitor, 24% alone), while 34% would start with IL-1/IL-6 in monotherapy. Factors favoring GCs use included severe pericarditis (64%), severe arthritis (55%), and refractory disease risk factors (16%). Biologics were used as 1st line by 76% of respondents, with anakinra being the most frequently used one (59%),followed by tocilizumab (25%). Factors driving anakinra’s choice were safety (72%), predominant systemic phenotype (73%) and cost (41%); while tocilizumab was chosen for arthritis-dominant profile (80%) and compliance (56%). Unavailability for Anakinra reached 17%, for Tocilizumab only 1.5%.In systemic-predominant SD, anakinra was the most frequently used medication (45%) in 1st line besides GCs and NSAIDs. In SD with predominant articular involvement, only half of clinicians would change the therapeutic approach, using more frequently TCZ, MTX and intra-articular steroids. Steroid tapering was guided mostly by clinical and laboratory parameters: acute phase reactants (95%) and ferritin (87%) reduction, fever resolution (82%, ≥1 week), arthritis improvement (75%).Methotrexate was the most common 1st-line choice for SD-refractory arthritis, followed by anti-TNF agents and intra-articular steroids. In SD patients with recurrent/refractory MAS, ciclosporin, anakinra, and MPN pulses were the most frequently selected medications. For SD-LD, 41% would continue ongoing biologics, 30% withdraw and 29% decide case by case. The most used 1st-line agents for SD-LD included JAK-i and mycophenolate mofetil. Most respondents (82%) would consider Hematopoietic Stem-Cell Transplantation (HSCT) in difficult-to-treat SD patients, particularly for refractory MAS or SD-LD.
Conclusion: Still’s disease still represents a therapeutic challenge, mainly due to its heterogeneity in clinical expression and limited evidences for refractory courses. Future research is essential to optimize clustering of patients to foster tailored target treatments, while ensuring equitable access to effective therapies worldwide.
Reasons affection biologic drugs choiche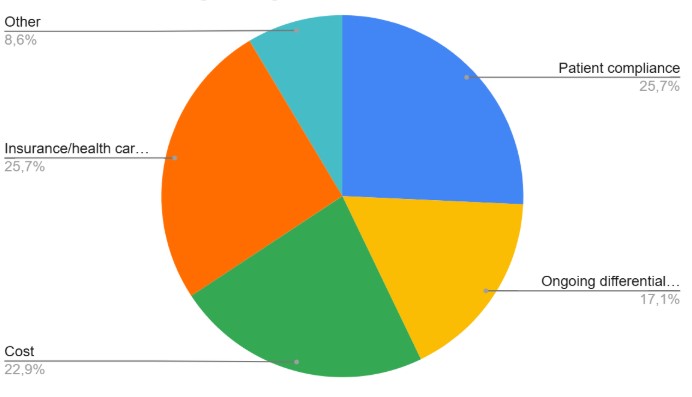
Top 4 preferred agents per refractory conditions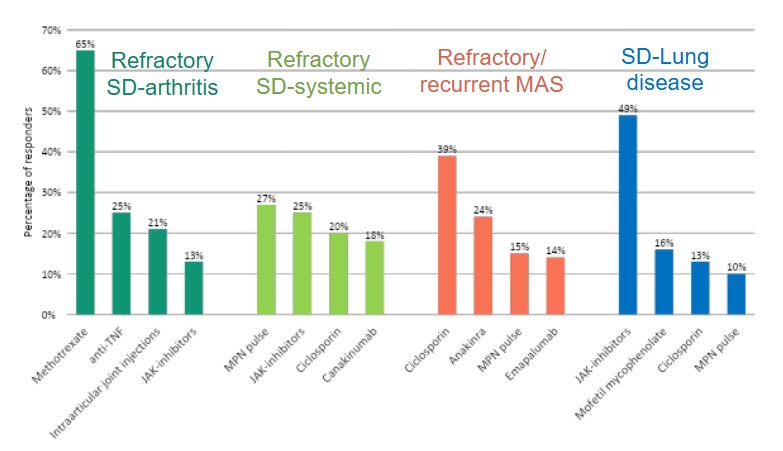
Different phenotypes, different therapy?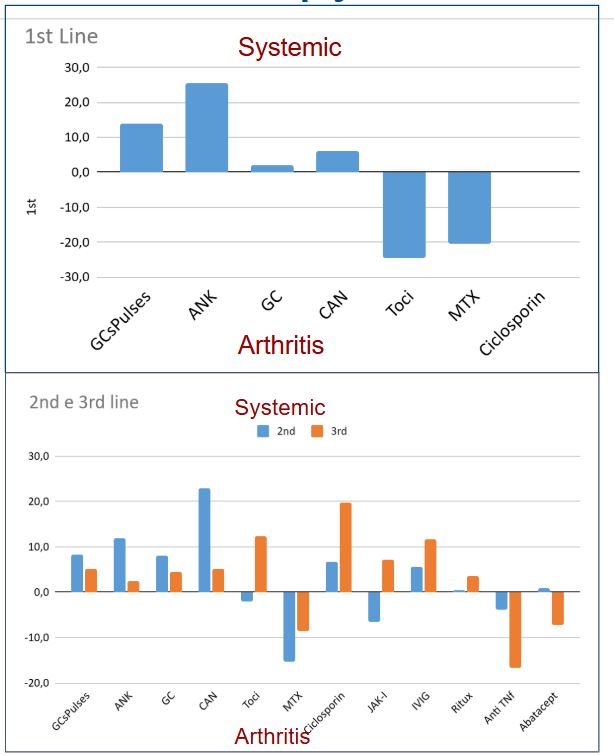
To cite this abstract in AMA style:
Baldo F, Rogani G, Bracaglia C, Foell D, Gattorno M, Anton J, Jelusic M, Brogan P, Canna S, Cron R, De Benedetti F, Grom A, Heshin Bekenstein M, Horne A, Khubchandani R, Mizuta M, Özen S, Quartier Dit Maire P, Ravelli A, Ruperto N, Shimizu M, Schulert G, Sinha R, Scott C, Swart J, Fautrel B, Vastert S, Minoia F. HOW DO WE HANDLE STILL’S DISEASE? REAL-LIFE CLINICIANS’ CHOICES FROM THE METAPHOR PROJECT WORLDWIDE SURVEY [abstract]. Arthritis Rheumatol. 2026; 78 (suppl 3). https://acrabstracts.org/abstract/how-do-we-handle-stills-disease-real-life-clinicians-choices-from-the-metaphor-project-worldwide-survey/. Accessed .« Back to 2026 Pediatric Rheumatology Symposium
ACR Meeting Abstracts - https://acrabstracts.org/abstract/how-do-we-handle-stills-disease-real-life-clinicians-choices-from-the-metaphor-project-worldwide-survey/
