Session Information
Session Time: 5:00PM-6:00PM
Background/Purpose: An increased risk of thromboembolic events among patients taking a janus kinase inhibitor (JAKi) was seen in The Oral Rheumatoid Arthritis Trial Surveillance study. This was a randomized, post authorization, noninferiority trial comparing the safety of tofacitinib to tumor necrosis factor inhibitors in rheumatoid arthritis. The results from this trial led the Food and Drug Administration to update drug labels for JAKi to include an increased risk of thrombosis. Subsequent studies have shown mixed and inconsistent results underscoring the need for additional investigation. Most studies to date are in adult populations and very limited data currently exists on the risk of thrombosis in pediatric patients on a JAKi. In this study we used the Epic Cosmos database to evaluate the relative risk of a thrombotic event in patients with juvenile idiopathic arthritis (JIA) treated with a JAKi compared to all patients with JIA.
Methods: Data for this analysis was extracted from Epic Cosmos, a dataset created in collaboration with a community of health systems using Epic representing more than 300 million patient records from over 1,800 hospitals and 41,500 clinics as of October 2025. The community represents patients from all 50 states, D.C., Canada, Lebanon, and Saudi Arabia. Like Epic’s SlicerDicer tool, users create a query by selecting population criteria and the outcome of interest. Our defined population included patients 21 years of age and younger with three sequential encounter diagnoses of JIA (International Classification of Diseases (ICD)-10 code M08.*) or psoriatic arthritis (L40.5), where the * indicates all codes under the umbrella code are included. The exposure group included patients with an active prescription for a JAKi in the study period (8/1/2015 – 7/31/2025), including upadacitinib, tofacitinib, ruxolitinib, baricitinib, abrocitinib, ritlecitinib, and fedratinib. The outcome events included pulmonary embolism (ICD-10 I26.*) and deep venous thrombosis (ICD-10 I80.*, I81.*, I82.*). Events within 5 years of JAKi initiation were considered. The Epic Cosmos platform calculates the risk ratio and associated 95% confidence interval.
Results: We identified 44,928 pediatric patients diagnosed with JIA during the 10-year study period (Table 1). The percentage of patients on a JAKi increased significantly during the study period from 0.3% to 11% (Figure). There were 46 thrombotic events in patients treated with a JAKi (Table 2). The corresponding risk ratio and 95% confidence interval were 1.399 [1.03,1.90].
Conclusion: This large population-level study suggests there is a slight trend towards increased risk of thrombotic events in JIA patients treated with JAKi compared to patients treated with other medications. Further analysis is needed to understand the role of other risk factors such as body mass index, medication dose, smoking status or exposure, pregnancy, systemic inflammation, comorbidities, and the impact of concomitant medications such as estrogen containing oral contraceptives. Epic’s Cosmos database may allow large volume observational studies in pediatric rheumatology and address prior research barriers associated with rare diseases.
Table 1: Characteristics of patients with JIA.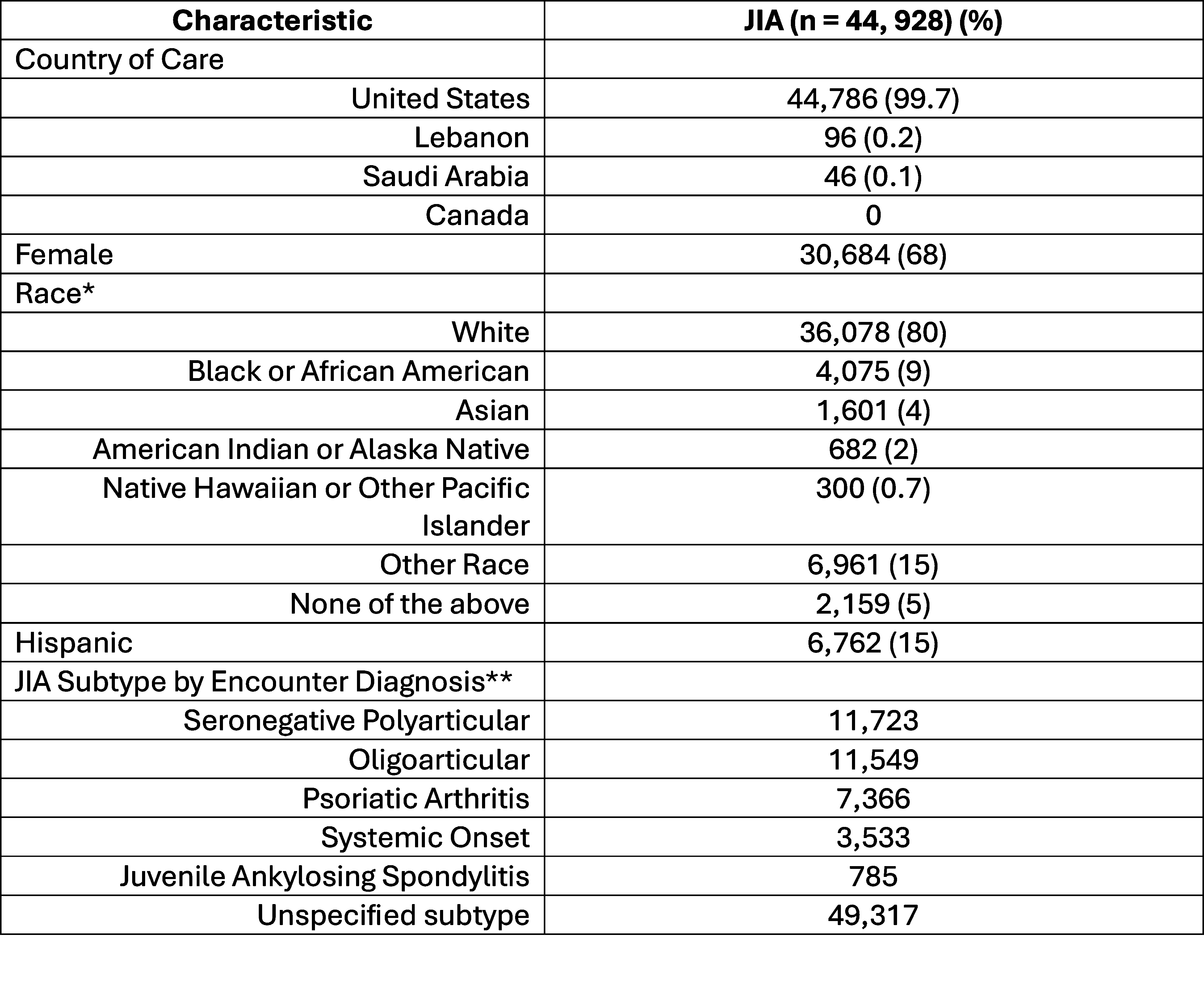 *A patient may have more than one race.
*A patient may have more than one race.
**Unspecified subtype includes ICD-10 codes with “unspecified” or “other” juvenile arthritis (for example, M08.80 = other juvenile arthritis). A patient may be included more than once in this category if each site of arthritis was included as a separate ICD-10 code.
Table 2: Risk of a thrombotic event in patients with a diagnosis of juvenile arthritis treated with JAKi. Risk ratio = 1.399 [1.03,1.90]
Risk ratio = 1.399 [1.03,1.90]
Figure. Percentage of patients treated with a JAKi over time.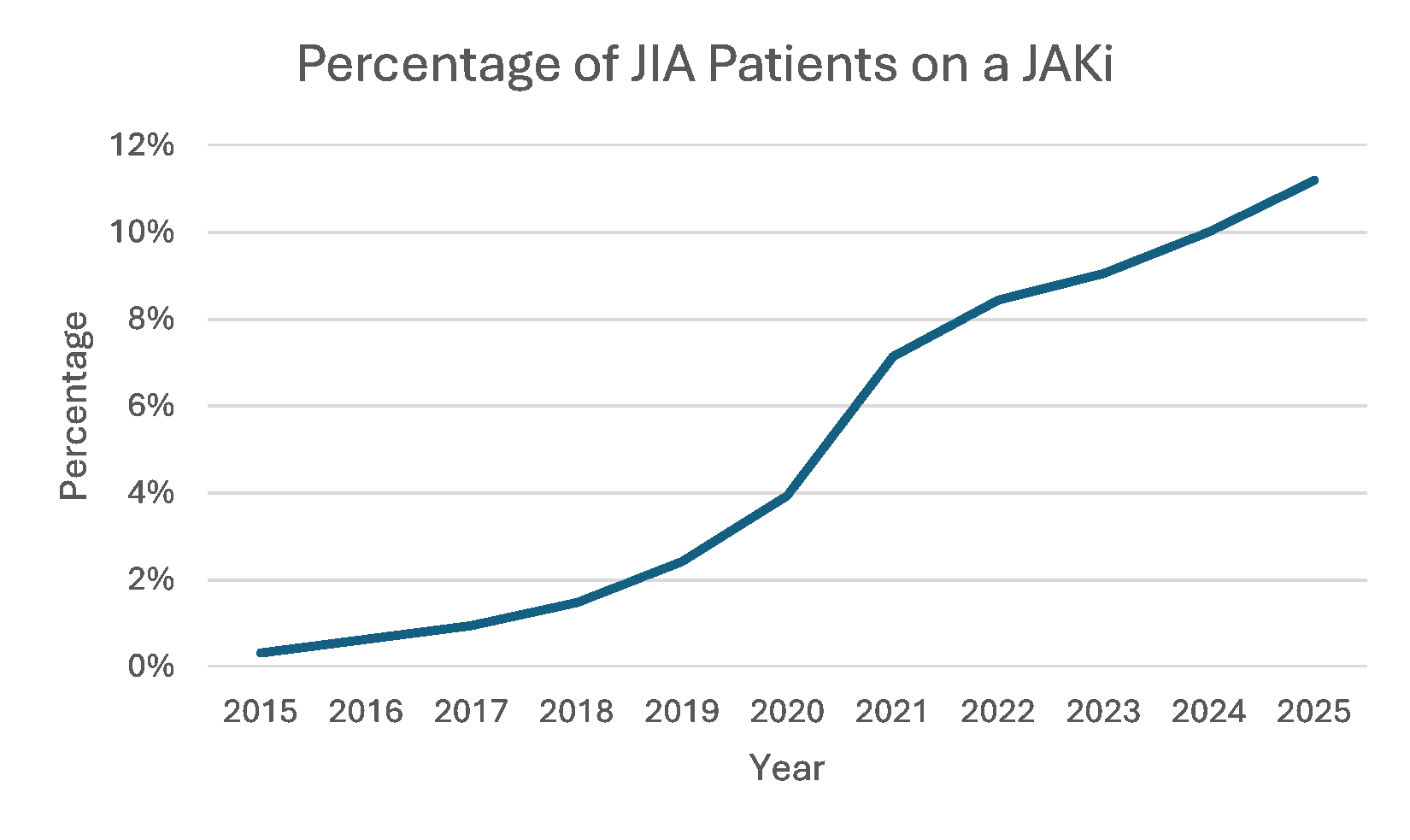
To cite this abstract in AMA style:
Taxter A, Dale M, Ronis T, Lara Castillo N, Pooni R, Rouster-Stevens K, Pinotti C, Clinical Informatics Workgroup C. Relative Risk of a Thrombotic Event in Janus Kinase Inhibitor Use for JIA using the Epic Cosmos Database [abstract]. Arthritis Rheumatol. 2026; 78 (suppl 3). https://acrabstracts.org/abstract/relative-risk-of-a-thrombotic-event-in-janus-kinase-inhibitor-use-for-jia-using-the-epic-cosmos-database/. Accessed .« Back to 2026 Pediatric Rheumatology Symposium
ACR Meeting Abstracts - https://acrabstracts.org/abstract/relative-risk-of-a-thrombotic-event-in-janus-kinase-inhibitor-use-for-jia-using-the-epic-cosmos-database/
