Session Information
Session Time: 5:00PM-6:00PM
Background/Purpose: Children with chronic, non-infectious uveitis (NIU) often require systemic, steroid-sparing therapy to control inflammation and prevent vision-threatening complications. Methotrexate (MTX) is the first-line systemic agent, yet 50-70% of children have an incomplete response, and therapeutic benefit can take three or more months to assess. Identifying clinical and demographic risk factors to predict MTX response could support earlier escalation to biologic medications. We aim to identify clinical and demographic predictors of inadequate response to MTX monotherapy in pediatric NIU patients.
Methods: This is a retrospective, single center, chart review study of patients diagnosed with NIU, seen between 2002-2022 and were treated with MTX. Ophthalmic risk factors of interest included, 1) uveitis laterality and location, 2) best-corrected visual acuity (BCVA) in the worse-seeing eye at diagnosis, 3) anterior cell grade at diagnosis and 6 weeks after therapy, 4) oral/topical steroid requirement in the first 6 weeks of diagnosis, and 5) total number of ocular complications. NIU patients were classified as responders to MTX monotherapy if 1) anterior chamber (AC) cell grade was classified as 0 in affected eye(s) and described as “quiet”, “quiescent”, “no active inflammation”, 2) absence of vitreous haze or other indications of uveitis activity by dilated exam/imaging, 3) ≤2 drops of prednisolone acetate 1% or equivalent/day, and 4) no oral corticosteroid. Clinical phenotypes and laboratory characteristics were compared between MTX inadequate responders (MTX-IR) and MTX responders (MTX-R) using Chi-square and two sample tests.
Results: A total of 51 patients met inclusion criteria. The cohort was predominantly White (82.4%) and female (62.8%), with 78.4% being identified as MTX-IR (Table 1). Over half of the population had systemic disease (50.9% JIU, 9.8% other), with 39.2% being diagnosed as undifferentiated NIU. On bivariate analysis, no statistical significance was identified between MTX-IR and demographic/clinical findings (race/ethnicity, sex, ESR, ANA, HLA-B27 status) or ophthalmic-specific risk-factors (Table 2). In 75% of patients who were identified as MTX-IR, the ANA was positive (p=0.0576), and while not statistically significant, had a trend towards significance.
Conclusion: Ultimately, we are unable to identify ophthalmic or other clinical risk factors that identify NIU patients at risk of MTX-IR. In contrast, other studies report severe eye inflammation and presence of complications at presentation to be associated with lack of response to MTX, though these findings are specifically in JIA-U. Our findings do not support evidence of using clinical criteria alone to identify at-risk populations for MTX-IR. Future investigation could include ocular findings on novel imaging modalities, biomarkers, and genetics as risk factors of MTX-IR, with the potential for developing individualized therapy plans while minimizing ophthalmic morbidity.
Table 1. Demographic and clinical characteristics associated with MTX response in patients with NIU 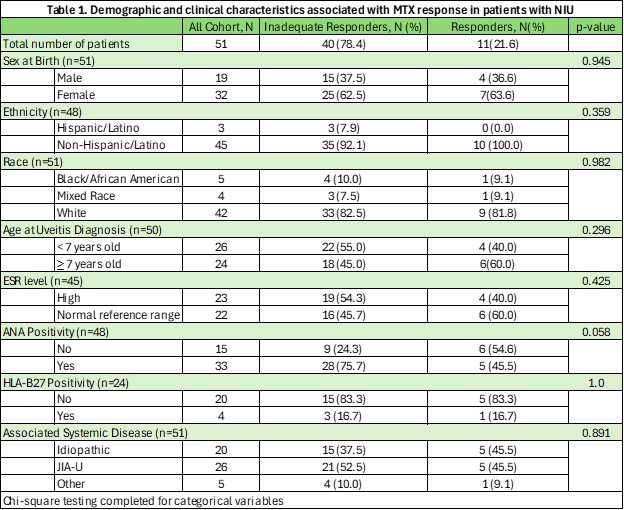
Table 2. Association between ophthalmological clinical characteristics and MTX response 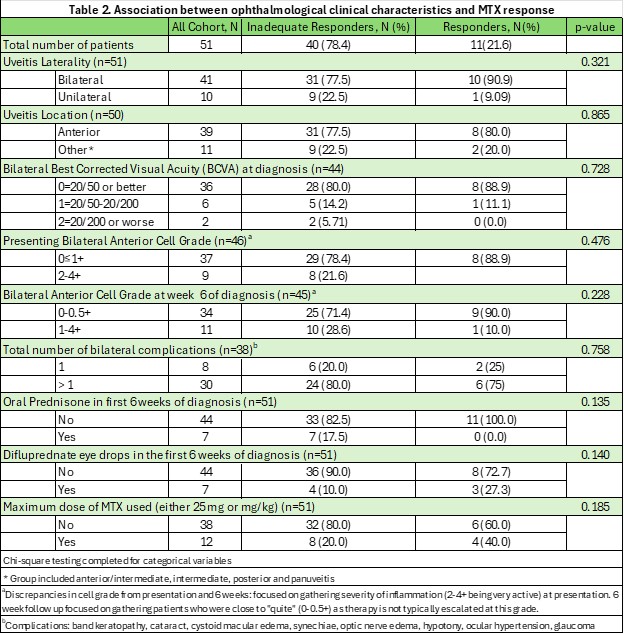
To cite this abstract in AMA style:
Jagger A, Asghar E, Quinlan-Waters M, Duell A, Altaye M, Miraldi Utz V, Angeles-Han S. Ophthalmic Predictors of Response to Methotrexate Monotherapy in Pediatric Non-Infectious Uveitis [abstract]. Arthritis Rheumatol. 2026; 78 (suppl 3). https://acrabstracts.org/abstract/ophthalmic-predictors-of-response-to-methotrexate-monotherapy-in-pediatric-non-infectious-uveitis/. Accessed .« Back to 2026 Pediatric Rheumatology Symposium
ACR Meeting Abstracts - https://acrabstracts.org/abstract/ophthalmic-predictors-of-response-to-methotrexate-monotherapy-in-pediatric-non-infectious-uveitis/
