Session Information
Session Time: 5:00PM-6:00PM
Background/Purpose: Therapeutic drug monitoring (TDM) can optimize patient outcomes in pediatric rheumatology, but its use is not standardized. This study aimed to explore TDM practice patterns and barriers among pediatric rheumatology providers in North America.
Methods: A survey was sent to 412 pediatric rheumatology providers in the United States and Canada, assessing practice patterns and barriers to use of TDM of TNF inhibitors (TNFi), azathioprine, cyclosporine, hydroxychloroquine, mycophenolate mofetil, and rituximab. Up to three reminder e-mails were sent to non-respondents. Respondents were categorized as “ever users” (i.e., those who had ever used TDM) or “never-users” of TDM for each medication. Ever users were further classified by frequency of TDM use: low-frequency (used in ≤25% of patients treated with the index medication in the last year) and high-frequency (used in >75% of patients treated with the index medication in the last year).
Results: There were 167 survey respondents (41%), primarily physicians (96%) working at academic centers (98%). TNFi had the highest proportion of ever-users of TDM (85%), while rituximab had the lowest (9%). Most medications had more low-frequency than high-frequency TDM users, except cyclosporine, for which high-frequency TDM use was common (n=61, 70%). The main barrier to TDM implementation was lack of training in use of TDM, followed by lack of scientific data in support of TDM in most cases. This held true across different levels of clinical experience.
Conclusion: This survey highlights marked variability in TDM use among pediatric rheumatology providers in North America. Future efforts should focus on clinical pharmacology education and expanding pharmacokinetics/pharmacodynamics studies to support incorporation of TDM into clinical guidelines, thereby improving personalized care and outcomes for children with rheumatic diseases.
Table 1. Survey Respondent and Practice-Related Characteristics‡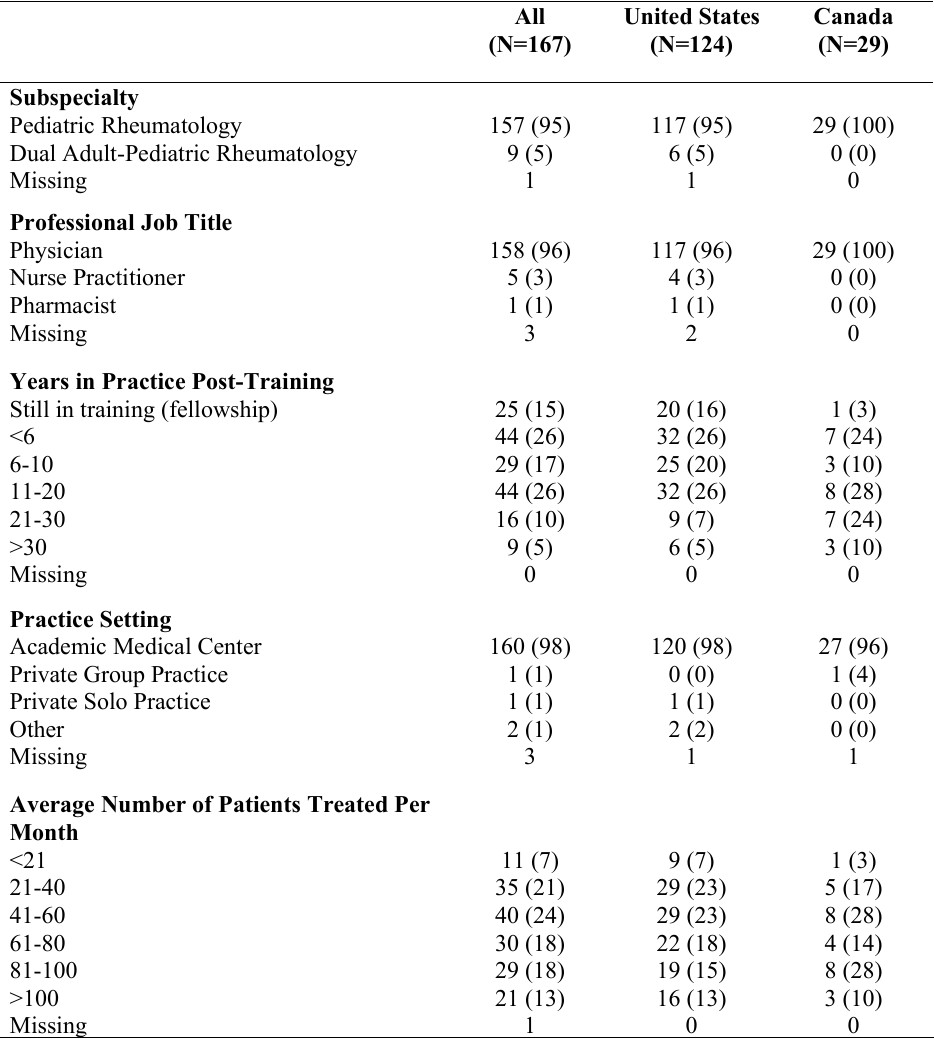 Legend. ‡All results expressed as n (%).
Legend. ‡All results expressed as n (%).
Table 2. TDM Practice Patterns‡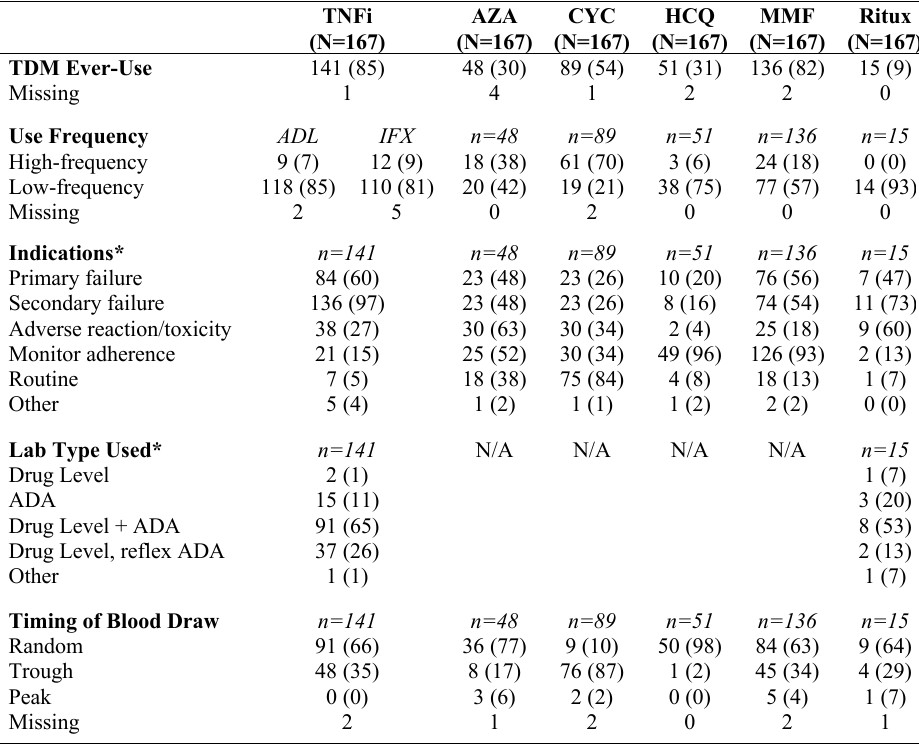 Legend. ‡All results expressed as n (%). Asterisks (*) indicate questions that allowed multiple responses. High-frequency use of TDM was defined as use in >75% of patients on a given medication in the last year. Low-frequency use was defined as use in ≤25% of patients on that medication in the last year. Primary failure was defined as lack of response to a medication from the outset. Secondary failure was defined as loss of response to a medication after a period of responsiveness. ADA = anti-drug antibodies; ADL = adalimumab; AZA = azathioprine; CYC = cyclosporine; HCQ = hydroxychloroquine; IFX = infliximab; MMF = mycophenolate mofetil; Ritux = rituximab; TNFi = TNF inhibitor.
Legend. ‡All results expressed as n (%). Asterisks (*) indicate questions that allowed multiple responses. High-frequency use of TDM was defined as use in >75% of patients on a given medication in the last year. Low-frequency use was defined as use in ≤25% of patients on that medication in the last year. Primary failure was defined as lack of response to a medication from the outset. Secondary failure was defined as loss of response to a medication after a period of responsiveness. ADA = anti-drug antibodies; ADL = adalimumab; AZA = azathioprine; CYC = cyclosporine; HCQ = hydroxychloroquine; IFX = infliximab; MMF = mycophenolate mofetil; Ritux = rituximab; TNFi = TNF inhibitor.
Figure. Percentage of survey respondents reporting each barrier to the use of TDM for each medication.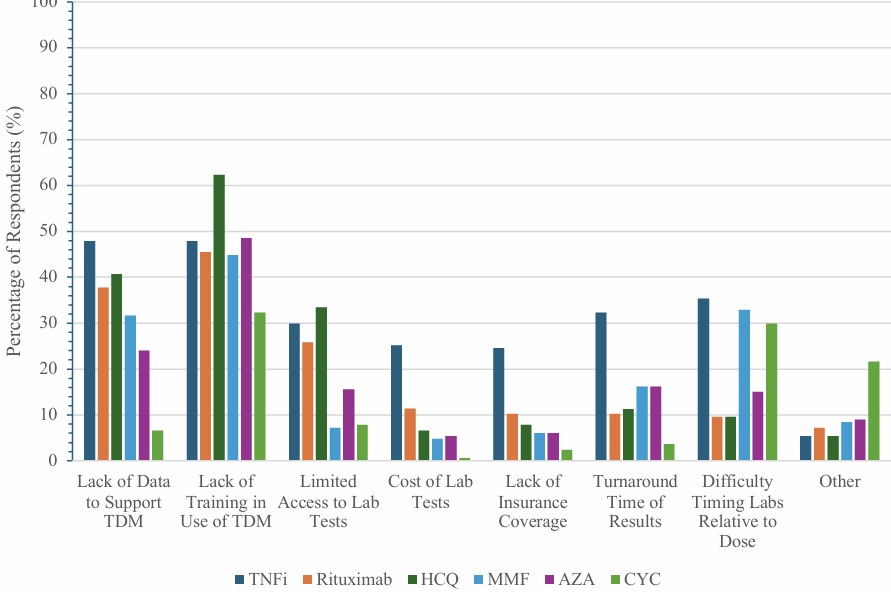 AZA = azathioprine; CYC = cyclosporine; HCQ = hydroxychloroquine; MMF = mycophenolate mofetil; TNFi = TNF inhibitor.
AZA = azathioprine; CYC = cyclosporine; HCQ = hydroxychloroquine; MMF = mycophenolate mofetil; TNFi = TNF inhibitor.
To cite this abstract in AMA style:
Sagcal-Gironella A, Randell R, Brunner H, Go E, Siu A, Verstegen R, DeGuzman M. Therapeutic Drug Monitoring in Pediatric Rheumatology: A Survey of Providers in North America [abstract]. Arthritis Rheumatol. 2026; 78 (suppl 3). https://acrabstracts.org/abstract/therapeutic-drug-monitoring-in-pediatric-rheumatology-a-survey-of-providers-in-north-america/. Accessed .« Back to 2026 Pediatric Rheumatology Symposium
ACR Meeting Abstracts - https://acrabstracts.org/abstract/therapeutic-drug-monitoring-in-pediatric-rheumatology-a-survey-of-providers-in-north-america/
