Session Information
Session Time: 6:00PM-7:00PM
Background/Purpose: Platelets are emerging as key modulators and protagonists of aberrant immune responses in severe systemic autoimmune diseases like systemic lupus erythematosus (SLE). While the hemostatic and thrombotic functions of platelets are better characterized, the pivotal interactions of platelets with the immune systems in systemic autoimmune diseases, and especially in pediatric-onset diseases, are still poorly understood. Studies on platelet activity, function, and genomics in childhood-onset SLE are greatly lacking. The objective of our study was to develop a high-parameter flow cytometry panel that will facilitate high-quality downstream multi-omics studies on thrombo-inflammation and cardiovascular disease in pediatric autoimmune disorders. We uniquely aimed to optimize our protocol to be suitable for small-volume whole blood sampling from younger pediatric patients.
Methods: Blood samples were collected from non-diseased individuals at Cincinnati Children’s Hospital Medical Center. Whole blood was collected into acid citrate dextrose (ACD) tubes. Complete blood count (leukocytes, erythrocytes, and platelets) was performed using an automatic hematology system. Platelet-rich plasma was prepared from centrifuged whole blood. Negative selection automated isolation was then performed after incubation of the platelet samples with microbead-conjugated antibodies against erythrocytes and leukocytes. Purification efficiency was verified with the automatic hematology system and by flow cytometry analysis pre- and post-isolation. Purified platelets were snap frozen and stored at -80 °C for subsequent omics analyses. For flow cytometry analysis, whole blood samples were diluted with Tyrode’s buffer and incubated with antibodies specific for leukocytes (CD45+), platelets (CD41+/CD42b+), and erythrocytes (RBC, CD235a+). Whole blood platelet activation status was analyzed with antibodies against activation markers aIIb-b3 (PAC-1), lysosomal activated membrane protein (CD63), p-selectin (CD62P), and inflammatory activation marker (membrane-bound CD40L).
Results: Eleven blood samples were analyzed. We found no difference in platelet yield and activation pattern comparing whole blood samples collected in ethylenediaminetetraacetic acid (EDTA) tubes versus ACD tubes (Figure 1). Our results also showed that platelet fixation with 1% PFA is required for optimal results of whole blood staining. We showed that blood fixation before staining is not needed. Flow cytometry results from pre- and post-automated platelet isolation showed absolute leukocyte and erythrocyte depletion. Our data also revealed no overall changes in platelet activation by our platelet isolation and purification process (Figure 2). We had sufficient platelet RNA yield which was reproducible. Using Bioanalyzer QC, we demonstrated that RNA extracted from the purified platelets was suitable for downstream multi-omics work.
Conclusion: Using rigorous techniques, our lab developed a 10-panel flow cytometry-based method to analyze platelet activation and function uniquely using a small amount of whole blood samples, which is key to accelerating platelet-based studies in pediatric autoimmune diseases.
FIGURE 1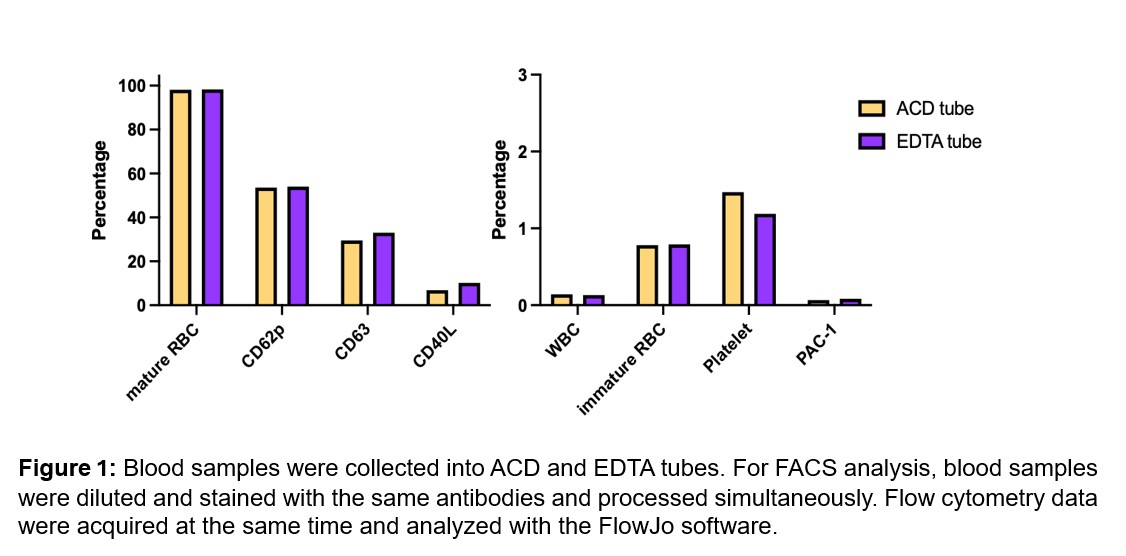 Figure 1: Blood samples were collected into ACD and EDTA tubes. For FACS analysis, blood samples were diluted and stained with the same antibodies and processed simultaneously. Flow cytometry data were acquired at the same time and analyzed with the FlowJo software.
Figure 1: Blood samples were collected into ACD and EDTA tubes. For FACS analysis, blood samples were diluted and stained with the same antibodies and processed simultaneously. Flow cytometry data were acquired at the same time and analyzed with the FlowJo software.
FIGURE 2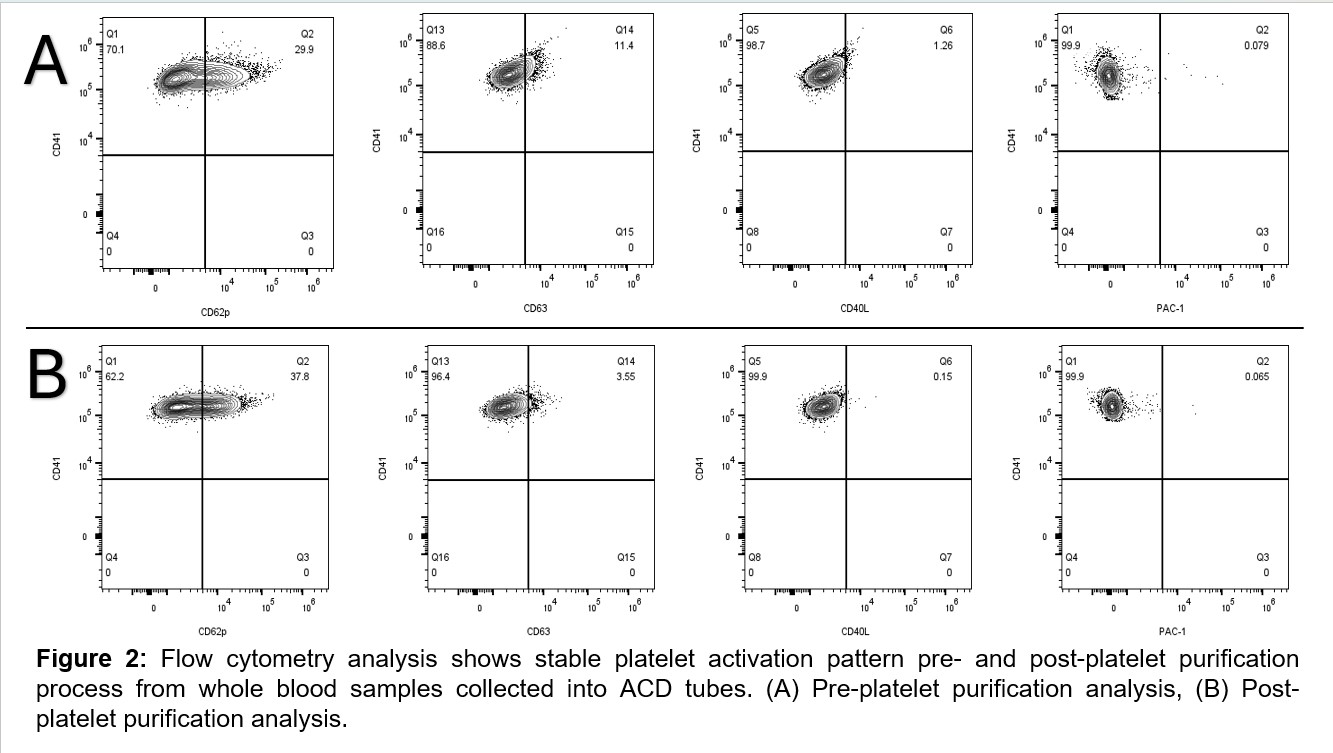 Figure 2: Flow cytometry analysis shows stable platelet activation pattern pre- and post-platelet purification process from whole blood samples collected into ACD tubes. (A) Pre-platelet purification analysis, (B) Post-platelet purification analysis.
Figure 2: Flow cytometry analysis shows stable platelet activation pattern pre- and post-platelet purification process from whole blood samples collected into ACD tubes. (A) Pre-platelet purification analysis, (B) Post-platelet purification analysis.
To cite this abstract in AMA style:
Zhen Y, Tourdot B, Zhang X, Lynch D, Ogbu E. Development of A High-Parameter Whole Blood Thrombo-inflammation Flow Cytometry Panel for Research in Pediatric Autoimmune Diseases [abstract]. Arthritis Rheumatol. 2026; 78 (suppl 3). https://acrabstracts.org/abstract/development-of-a-high-parameter-whole-blood-thrombo-inflammation-flow-cytometry-panel-for-research-in-pediatric-autoimmune-diseases/. Accessed .« Back to 2026 Pediatric Rheumatology Symposium
ACR Meeting Abstracts - https://acrabstracts.org/abstract/development-of-a-high-parameter-whole-blood-thrombo-inflammation-flow-cytometry-panel-for-research-in-pediatric-autoimmune-diseases/
