Session Information
Session Time: 6:00PM-7:00PM
Background/Purpose: Many children with juvenile spondyloarthritis (JSpA) express interest in discontinuing medication once inactive disease is achieved. The BACK-OFF JSpA Trial evaluates flare risk and patient experiences across TNFi de-escalation strategies. This report summarizes the impact of stakeholder-designed recruitment materials, the baseline characteristics of enrolled participants, and their preferences for sharing study results once available.
Methods: BACK-OFF JSpA trial enrolled children with JSpA on TNFi therapy and sustained inactive disease for ≥6 months. Participants were randomized to continue standard TNFi dosing, extend the TNFi dosing interval, or discontinue TNFi therapy. Trial participation lasts 12 months or until a protocol-defined disease flare occurs, whichever comes first. Patient and parent stakeholder partners collaborated on the study design and creation of recruitment materials. Participants were asked how they would prefer to receive information about the study results once they became available, via a REDCap questionnaire.
Results: Enrollment in the BACK-OFF JSpA trial is now complete, with 164 participants enrolled across 31 sites. All participating sites enrolled at least one participant, and over half enrolled four or more. 52% of participants were enrolled at the first approach. Among enrolled participants, 64% acknowledged reviewing stakeholder-developed recruitment materials before or at the time of enrollment (Table 1). 66% rated the impact of these materials as positive, with 29% and 27% indicating that the materials influenced their decision to enroll “a little” or “a lot”, respectively. Table 2 shows that the baseline cohort consisted of 71% males, with a median age of 16.5 years; 56% of the cohort were HLA-B27 positive. At enrollment, the median disease duration was 30 months, and the median duration of inactive disease was 12.2 months. 15% of participants previously attempted TNFi de-escalation. 88% of participants expressed interest in hearing about the study results as soon as they became available. Participant preferences for results-sharing formats were as follows: website (38%), scientific paper (27%), pamphlet or flyer (17%), webinar (10%), podcast (5%), and other (3%) (Figure 1). 53% of the respondents reported no preference regarding who should communicate the results, while 39%, 6%, and 2% preferred to hear about the results from the local rheumatology team, the core study team, or the stakeholder panel, respectively.
Conclusion: Enrollment in the BACK-OFF JSpA trial is complete, with strong engagement across sites. Stakeholder-developed recruitment materials were well-received and influenced enrollment decisions for many participants, supporting the value of stakeholder partner involvement in trial design. Most participants expressed interest in timely access to study results, favoring digital formats such as websites and scientific papers, with no strong preference for who shared the results. These findings underscore the importance of stakeholder-partner-informed communication strategies in enhancing engagement throughout the research process.
Table 1: Efficacy of Recruitment Materials Legend. ^Missing for 5 enrolled subjects
Legend. ^Missing for 5 enrolled subjects
Table 2. Participants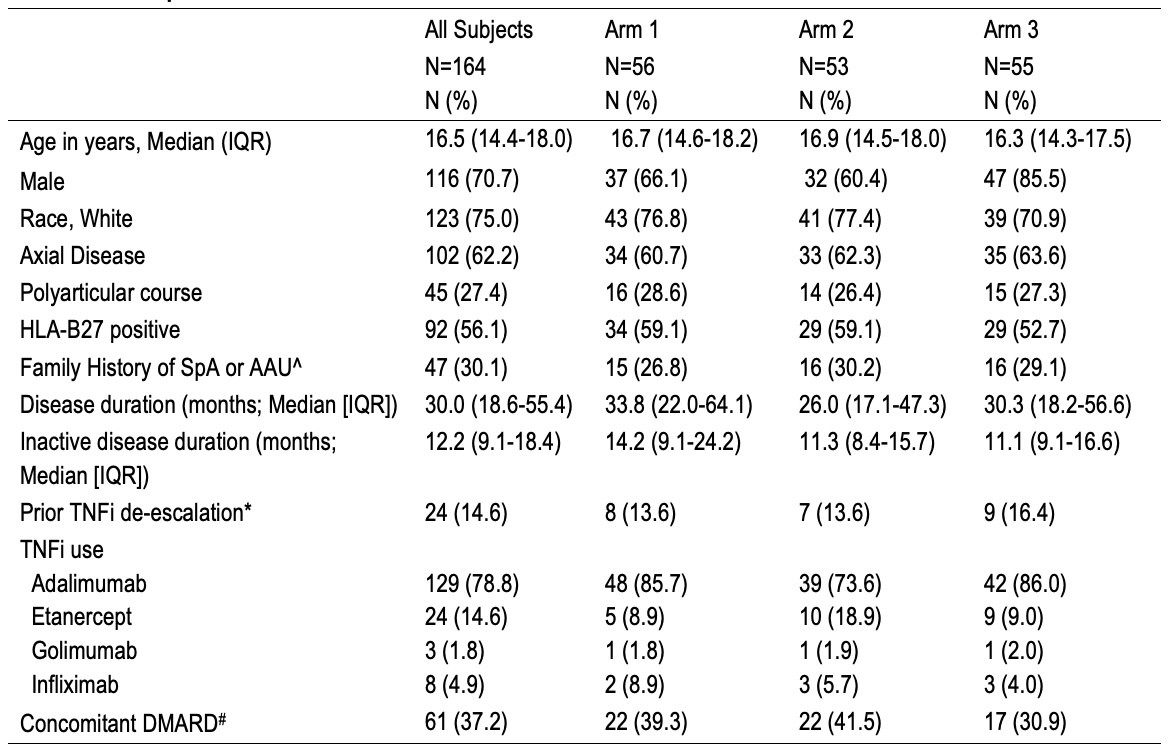 Legend. Participants randomized to one of 3 arms at enrollment- continuation of standard dosing, less frequent dosing, or stopping TNFi. ^Acute anterior uveitis. #Disease-modifying antirheumatic drug which includes methotrexate, sulfasalazine, or leflunomide. * TNFi de-escalation included reducing the dose, change in dose frequency, or stopping drug.
Legend. Participants randomized to one of 3 arms at enrollment- continuation of standard dosing, less frequent dosing, or stopping TNFi. ^Acute anterior uveitis. #Disease-modifying antirheumatic drug which includes methotrexate, sulfasalazine, or leflunomide. * TNFi de-escalation included reducing the dose, change in dose frequency, or stopping drug.
Figure 1. Dissemination Preferences of Participants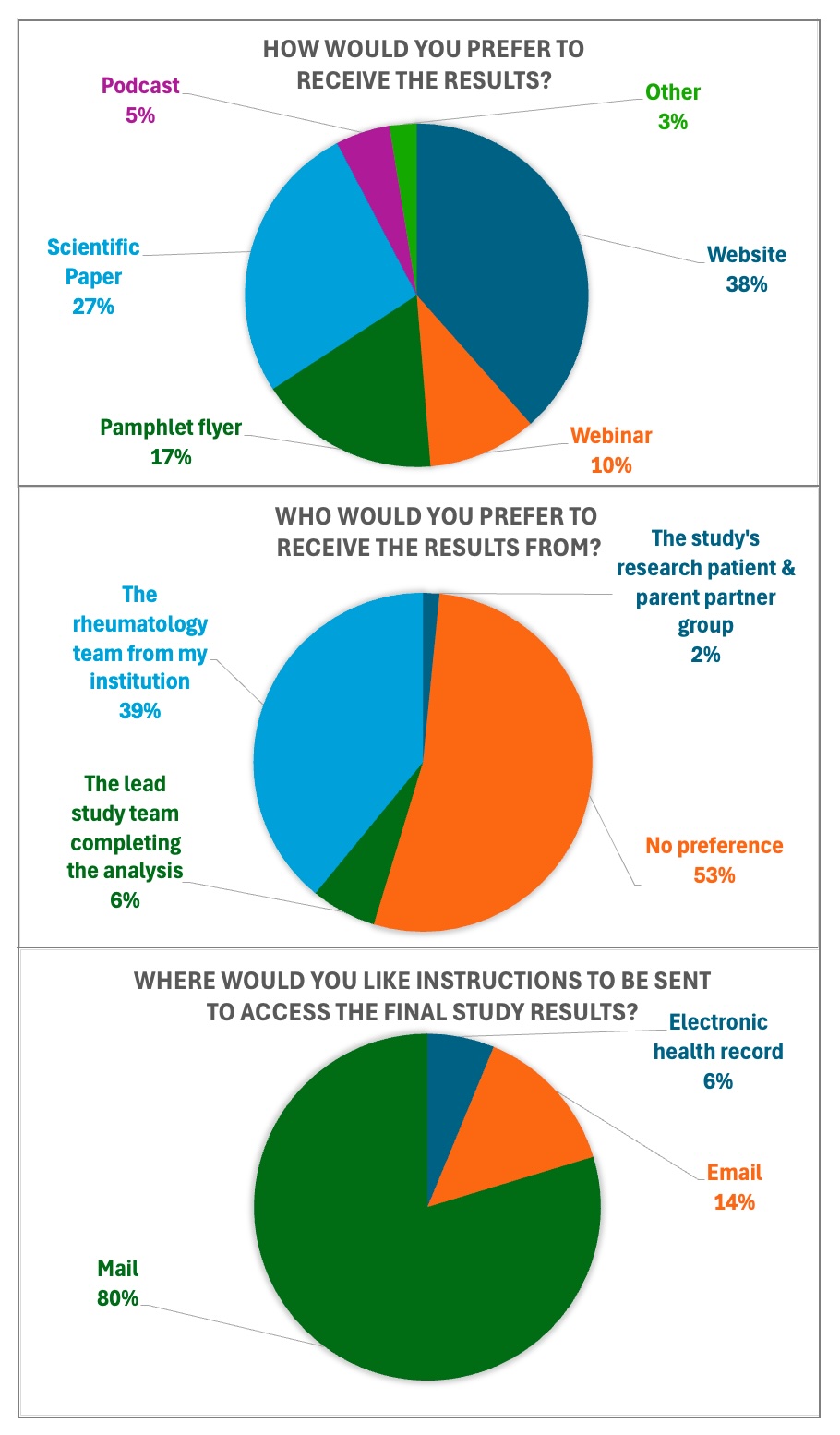 Nf 64 participants.
Nf 64 participants.
To cite this abstract in AMA style:
Sears C, Baradziej K, Brandon T, Ferguson P, Correll C, Rosenkranz M, Baszis K, Lee T, oberle E, Stoll M, Cook K, Muscal E, Sule S, Lovell D, Moore K, Prahalad S, Cidon M, Mulvihill E, Klein-Gitelman M, Kingsbury D, Rosenwasser N, Treemarcki E, Tarvin S, Chang J, Walters H, Shishov M, Buckley L, Toth M, cooper a, Wright T, Tse S, Bilgic Dagci A, Onel K, Xiao R, Neu E, Kohlheim M, Leal J, Archie K, Holland E, Holland M, Hameed A, Khan A, Murphy L, Murphy S, Neu J, Suplee D, Suplee T, Wiley D, Weiss P. The Biologic Abatement and Capturing Kids’ Outcomes and Flare Frequency in Juvenile Spondyloarthritis trial: Baseline cohort characteristics and importance of stakeholder partner-informed knowledge translation and exchange [abstract]. Arthritis Rheumatol. 2026; 78 (suppl 3). https://acrabstracts.org/abstract/the-biologic-abatement-and-capturing-kids-outcomes-and-flare-frequency-in-juvenile-spondyloarthritis-trial-baseline-cohort-characteristics-and-importance-of-stakeholder-partner-informed-kno/. Accessed .« Back to 2026 Pediatric Rheumatology Symposium
ACR Meeting Abstracts - https://acrabstracts.org/abstract/the-biologic-abatement-and-capturing-kids-outcomes-and-flare-frequency-in-juvenile-spondyloarthritis-trial-baseline-cohort-characteristics-and-importance-of-stakeholder-partner-informed-kno/
