Session Information
Session Time: 6:00PM-7:00PM
Background/Purpose: Antineutrophil cytoplasmic antibody-associated vasculitis (AAV) is a severe small vessel vasculitis. Rituximab is a monoclonal anti-CD20 antibody that is effective for induction and maintenance of remission in AAV. Guidelines recommend using rituximab for induction of severe AAV. In pediatric populations, rituximab has demonstrated comparable safety and efficacy to adults. However, data regarding the use of rituximab biosimilars in pediatric AAV remain scarce, with most available evidence derived from adult studies and regulatory extrapolation. Our aim is to report our experience in using rituximab biosimilars in pediatric AAV.
Methods: This is a retrospective, single-center retrospective review of pediatric AAV patients who were treated with rituximab biosimilars between 1/1/2020 and 8/1/2025. Data were obtained from the electronic medical record review.
Results: Eight pediatric patients with granulomatosis with polyangiitis (GPA) were included. The average age of onset was 12 years (range 7–16), and the average age at diagnosis was 12.13 years (range 7–16). Mean follow-up duration was 30.44 months (range 3.7–49.7). Of the cohort, five were female and three were male.Most patients had GPA (7/8) and one had MPA. The average time from first reported symptom to induction with rituximab was 2.31 months (range 0.4–4.2). For induction, Truxema was used 7/8; one of whom had a single dose of Rituxan before switching to Truxema. Ruxience was used in two patients. No patients failed to achieve CD20+ B-cell depletion following induction.Hypogammaglobulinemia with IgG levels < 400 mg/dL occurred at some point in 6 out of 8 patients. Two recovered within 3 months of induction; one recovered at 7.3 months after induction; one recovered 15.8 months post induction; and two had persistent hypogammaglobulinemia.ANCA titers became negative in 4/8 patients at 2.6, 4.6, 4.9, and 5.6 months post induction. One patient with shorter follow-up had improved but not negative titers. One seroconverted well into maintenance phase (10.2 months), and one patient had persistent positive titers.Maintenance therapy was initiated in 7/8 patients. Five received rituximab for maintenance, all with Ruxience. Of these, one developed COVID-19 and serum sickness, prompting a switch to azathioprine. Another transitioned to azathioprine after two years of maintenance rituximab but experienced worsening disease and undetectable serum azathioprine levels; disease was recaptured on resuming rituximab. One patient had worsening disease and was switched to oral cyclophosphamide followed by azathioprine. One patient opted for maintenance azathioprine after rituximab induction. One patient has not yet started maintenance therapy.Two experienced end-stage renal disease and are dialysis dependent. No patients died. Other complications included subglottic stenosis and deep vein thrombosis.
Conclusion: Our experience shows that rituximab biosimilars can be effective for pediatric AAV patients. We are unable to assess the relative safety and efficacy, and further study is needed to confirm.
Table 1: Case Series Table of Participants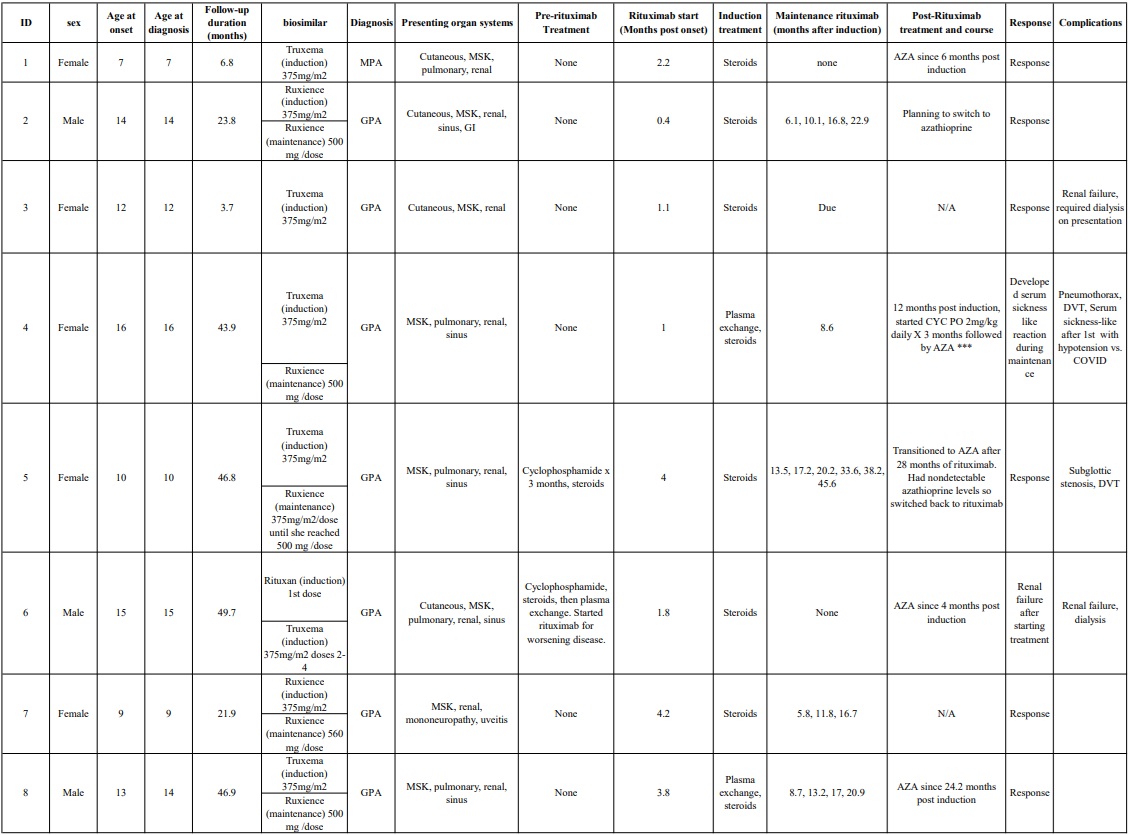
To cite this abstract in AMA style:
Mitacek R, Valentini R, Baracco R, Fathalla B. Efficacy of Rituximab Biosimilars in Pediatric ANCA-Associated Vasculitis [abstract]. Arthritis Rheumatol. 2026; 78 (suppl 3). https://acrabstracts.org/abstract/efficacy-of-rituximab-biosimilars-in-pediatric-anca-associated-vasculitis/. Accessed .« Back to 2026 Pediatric Rheumatology Symposium
ACR Meeting Abstracts - https://acrabstracts.org/abstract/efficacy-of-rituximab-biosimilars-in-pediatric-anca-associated-vasculitis/
