Session Information
Session Time: 6:00PM-7:00PM
Background/Purpose: Systemic lupus erythematosus (SLE) is a heterogeneous disease, and pediatric lupus nephritis (LN) is a severe manifestation affecting 50-60% of children with SLE, contributing significantly to morbidity and mortality. Standard induction therapy involves corticosteroids and an immunosuppressive agent like mycophenolate mofetil (MMF). Recent guidelines support adding a calcineurin inhibitor to the management of LN. Voclosporin, a novel calcineurin inhibitor, is approved for adult LN and has demonstrated improved renal response rates in clinical trials when added to standard therapy. With a favorable pharmacokinetic profile that does not require therapeutic drug monitoring, it is an attractive option for pediatrics. Given that pediatric patients were underrepresented in pivotal adult trials, this study describes the single-center experience and outcomes of using voclosporin for pediatric patients with refractory or relapsed LN.
Methods: A retrospective chart review was conducted for a cohort of five pediatric patients, all of whom had SLE according to ACR classification criteria, who received off-label voclosporin as part of their LN treatment. Patients had biopsy-proven LN of varying classes (IV, V, and IV/V), and had previously failed or relapsed on multiple therapeutic approaches. Voclosporin was initiated between 11 and 23 months after the LN diagnosis. The primary outcome measure was the change in the urine protein-to-creatinine ratio (Pr/Cr) following initiation of voclosporin. All patients were diagnosed and managed in accordance with current international classification and treatment guidelines for SLE and LN.
Results: Five pediatric patients (aged 10-15 years at SLE diagnosis) with refractory or relapsed LN were treated with voclosporin (doses of 15.8-23.7 mg twice daily). One patient with Class IV/V LN, after failing multiple agents, achieved significant improvement in proteinuria and sustained renal remission without side effects . Another patient who had persistent proteinuria despite MMF, tacrolimus, and intravenous cyclophosphamide achieved a substantial reduction in proteinuria, which allowed for steroid discontinuation and sustained remission. A third patient with Class V LN who had persistent renal involvement despite MMF, hydroxychloroquine, and belimumab experienced rapid improvement in proteinuria after voclosporin was added. One patient discontinued voclosporin after 14 months of good renal response due to persistent hypertension. Overall, the treatment was associated with a reduction in the mean urine Protein/Creatinine ratio across the cohort.
Conclusion: Voclosporin appears to be a safe and effective treatment option for pediatric patients with lupus nephritis, particularly in cases of refractory or relapsed disease. The reduction in proteinuria and overall sustained remission observed in this small cohort support the ongoing investigation into this agent for the management of severe pediatric LN.
Figure 1- patients respose to treatment with voclosporin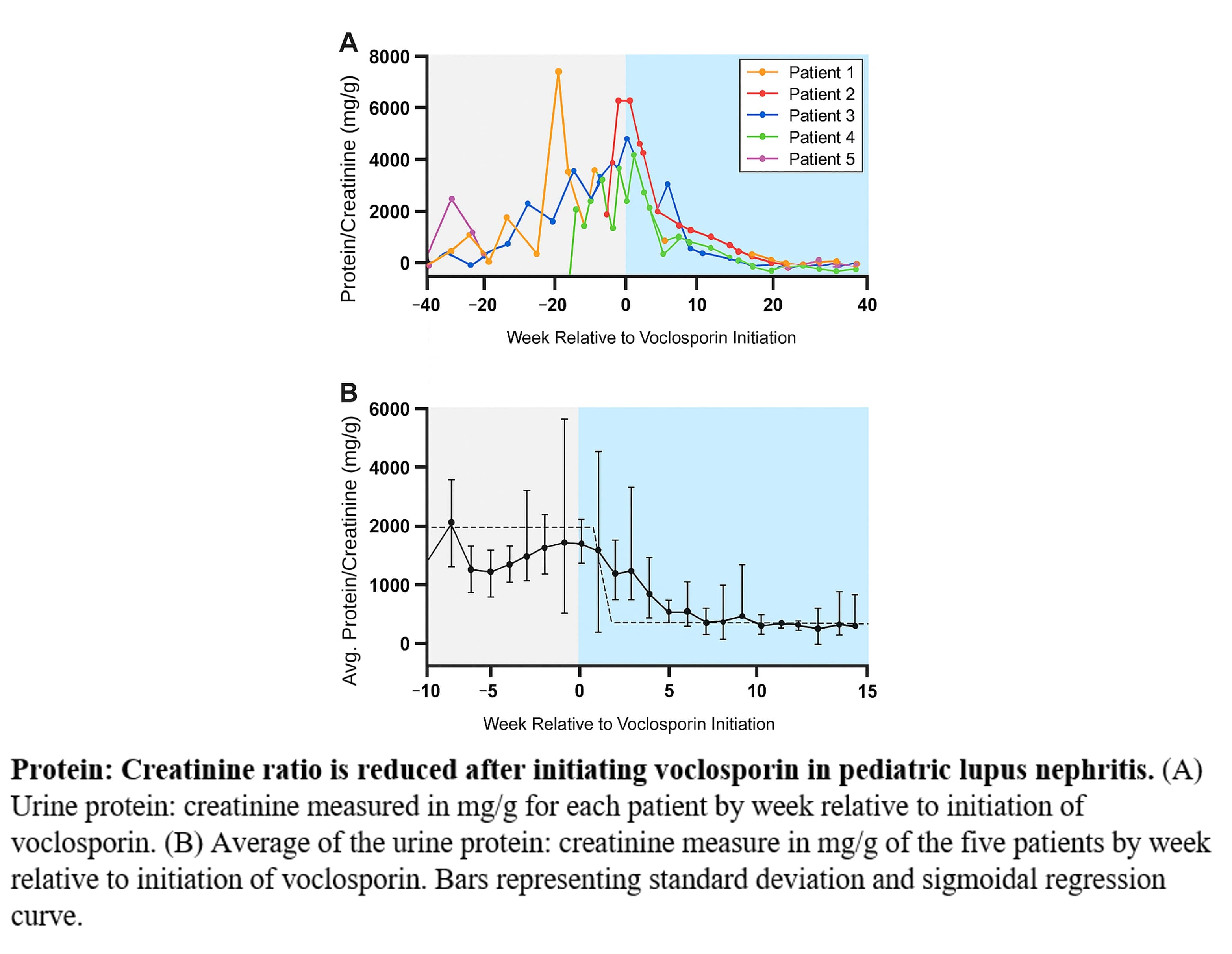 protein/creatinin ratio in response to voclosporin treatment initiation
protein/creatinin ratio in response to voclosporin treatment initiation
Table 1- patients’ characteristics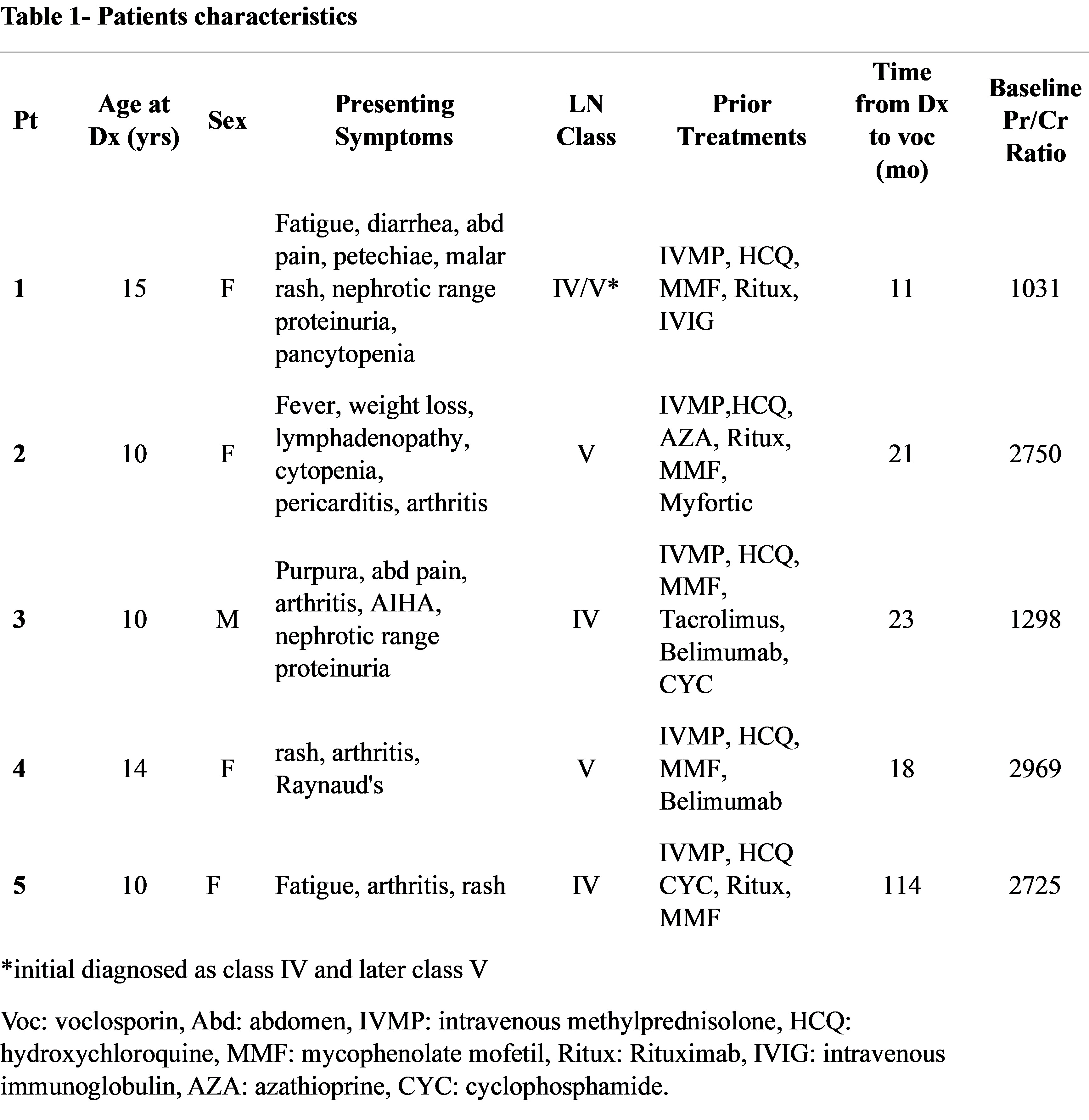 Table 1- patients’ characteristics
Table 1- patients’ characteristics
To cite this abstract in AMA style:
Hazan R, Civilibal N, Baszis K, Bigley T. Single-center Experience of Voclosporin Use for Pediatric Lupus Nephritis [abstract]. Arthritis Rheumatol. 2026; 78 (suppl 3). https://acrabstracts.org/abstract/single-center-experience-of-voclosporin-use-for-pediatric-lupus-nephritis/. Accessed .« Back to 2026 Pediatric Rheumatology Symposium
ACR Meeting Abstracts - https://acrabstracts.org/abstract/single-center-experience-of-voclosporin-use-for-pediatric-lupus-nephritis/
