Session Information
Session Time: 6:00PM-7:00PM
Background/Purpose: Childhood-onset systemic lupus erythematosus (SLE) is typically more severe than adult-onset disease, with higher morbidity, organ damage, and mortality. Despite this, pediatric drug trials lag behind adult studies and current treatment remains largely informed by adult data. Chimeric antigen receptor (CAR) T-cell therapy shows promise in autoimmune (AI) diseases in adults, and there is a growing interest in exploring its use in pediatric AI diseases; however, autologous CAR T-cell approaches are limited by prolonged pre/post-apheresis timelines, restricted access to authorized treatment centers, T-cell product inconsistency, high costs, and production capacity constraints. FT819 is an off-the-shelf iPSC-derived CD19-targeting CAR T-cell product candidate with a 1XX CAR signaling domain; integration of CAR transgene directly into the TCR alpha constant locus; and complete disruption of TCR expression for prevention of GvHD. FT819 is under study in an ongoing Phase 1, open-label, dose-escalation trial (NCT06308978) evaluating the safety, efficacy, pharmacokinetics, and anti-B cell activity of FT819 in patients (pts) with B-cell mediated AI diseases: SLE, AAV, IIM, and SSc. To accelerate pediatric access to FT819, this study is now enrolling pts aged ≥12 years. Here, we describe the study design and present initial data supporting enrollment of adolescent pts.
Methods: This study consists of a 3+3 dose-escalation phase, followed by cohort expansion. Eligible SLE pts met EULAR/ACR 2019 classification with at least 1 antibody (ANA ≥1:160, anti-dsDNA, or anti-Smith) and had SLEDAI-2K ≥8 plus BILAG 1A/2B scores. Pts received a single dose of FT819 under one of two regimens (Reg): Reg A, discontinuation of immunosuppression followed by fludarabine-free conditioning chemotherapy (CCT) with single-dose cyclophosphamide or bendamustine daily for 2 days prior to FT819; or Reg B, no CCT with continued stable maintenance therapy, including mycophenolate (Figure 1). Hydroxychloroquine (HCQ) and ≤10 mg prednisone (GC) equivalent were permitted. Within each regimen, pts received either dose level (DL) 1 (3.6 x 108 viable cells [VC]/dose) or DL2 (9 x 108 VC/dose) per the dose-escalation schedule.
Results: As of October 22, 2025, 10 enrolled adult pts completed ≥1 month (M) follow-up after receiving FT819 (Reg A, n=8; Reg B, n=2). All had SLE; baseline (BL) characteristics are shown in Table 1. Figure 2 shows a mean reduction in SLEDAI-2K score of 12.0±4.3 at 6M post-FT819 (n=4), along with decreases in UPCr over time and improvements in PGA and FACIT-Fatigue scores.
Of the 7 pts on GC at BL, 3 discontinued GC; 3 are on ≤5 mg/day. No Grade >2 CRS, ICANS, GvHD, or deaths were reported on study. All pts tolerated FT819 without dose-limiting toxicity. B-cell depletion was seen with reconstitution towards predominantly naïve cells by 1-3M. Updated data will be presented.
Conclusion: Preliminary data in pts with moderate-to-severe SLE treated on-demand with FT819 with less-intensive or no CCT demonstrate a favorable tolerability profile, effective B-cell depletion, and encouraging efficacy, and led to opening enrollment for adolescent patients.
Figure 1. FT819-102 Study Treatment Schema and Therapeutic Approach AAV = ANCA-associated vasculitis; AZA = azathioprine; DLT = dose-limiting toxicity; IIM = idiopathic inflammatory myopathies; LTFU = long-term follow-up; MMF/MPA = mycophenolate mofetil/mycophenolic acid; MTX = methotrexate; PTFU = post-treatment follow-up; SLE = systemic lupus erythematosus; SSc = systemic sclerosis.
AAV = ANCA-associated vasculitis; AZA = azathioprine; DLT = dose-limiting toxicity; IIM = idiopathic inflammatory myopathies; LTFU = long-term follow-up; MMF/MPA = mycophenolate mofetil/mycophenolic acid; MTX = methotrexate; PTFU = post-treatment follow-up; SLE = systemic lupus erythematosus; SSc = systemic sclerosis.
Table 1. Baseline Characteristics of SLE Patients Treated with FT819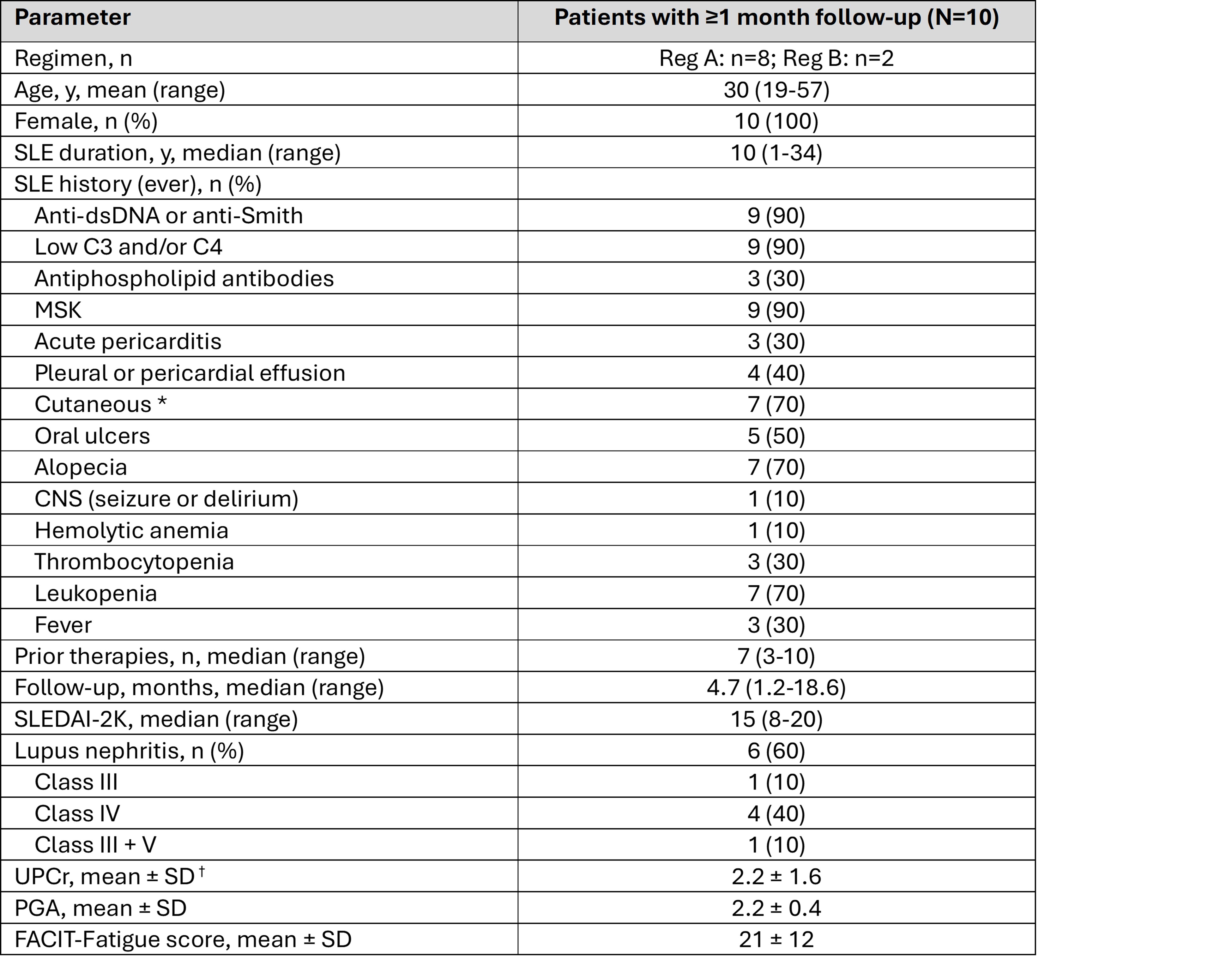 * Includes subacute, discoid, and/or acute cutaneous lupus.
* Includes subacute, discoid, and/or acute cutaneous lupus.
† Measured in patients with lupus nephritis.
All patients had previously received glucocorticoids (GC) and hydroxychloroquine (HCQ).
CNS = central nervous system; FACIT = Functional Assessment of Chronic Illness Therapy; MSK = musculoskeletal; PGA = physician’s global assessment; SLE = systemic lupus erythematosus; SLEDAI-2K = Systemic Lupus Erythematosus Disease Activity Index 2000; UPCr = urine protein-to-creatinine ratio.
Figure 2. SLEDAI-2K, PGA, UPCr, and FACIT-Fatigue Assessed at Baseline (Pre-FT819) and Study Time Points  One patient resumed mycophenolate at ~7.5 months having previously received this therapy for >4 years prior to CAR T-cell therapy. Another patient (without lupus nephritis) resumed anifrolumab at ~2 months, having previously received this therapy for 3 years prior to CAR T-cell therapy. One patient discontinued the study after the 1-month visit due to inability to meet study requirements.
One patient resumed mycophenolate at ~7.5 months having previously received this therapy for >4 years prior to CAR T-cell therapy. Another patient (without lupus nephritis) resumed anifrolumab at ~2 months, having previously received this therapy for 3 years prior to CAR T-cell therapy. One patient discontinued the study after the 1-month visit due to inability to meet study requirements.
To cite this abstract in AMA style:
Shiff N, Medlin J, Fazeli P, Pang S, Desai S, BitMansour A, Bethune C, Zack D, Elstrom R, Greene T, Wong L, Wong C, Ferguson B, Sandhu V. Off-the-Shelf Anti-CD19 CAR T-cell Therapy with Reduced or No Conditioning Chemotherapy Demonstrates Efficacy and Tolerability in Systemic Lupus Erythematosus: Meaningful Activity Supports Planned Adolescent Enrollment [abstract]. Arthritis Rheumatol. 2026; 78 (suppl 3). https://acrabstracts.org/abstract/off-the-shelf-anti-cd19-car-t-cell-therapy-with-reduced-or-no-conditioning-chemotherapy-demonstrates-efficacy-and-tolerability-in-systemic-lupus-erythematosus-meaningful-activity-supports-planned-ado/. Accessed .« Back to 2026 Pediatric Rheumatology Symposium
ACR Meeting Abstracts - https://acrabstracts.org/abstract/off-the-shelf-anti-cd19-car-t-cell-therapy-with-reduced-or-no-conditioning-chemotherapy-demonstrates-efficacy-and-tolerability-in-systemic-lupus-erythematosus-meaningful-activity-supports-planned-ado/
