Session Information
Session Time: 5:35PM-5:40PM
Background/Purpose: We assessed patterns of healthcare and medication utilization in children newly diagnosed with JIA compared to matched children without JIA. We hypothesized that healthcare utilization and use of anti-inflammatory and pain medications would be higher for JIA than matched controls, both before, around, and after diagnosis.
Methods: Using national Medicaid insurance data from 2001-2019, we identified children with newly diagnosed JIA (cases) based on recently validated algorithms who were continuously enrolled for ≥3 years before diagnosis until ≥1 year post-diagnosis. Each case was matched at the diagnosis (index) date to 1-10 controls with no prior JIA or immunosuppressant use by birth quarter, sex, state, and age at enrollment. Anchored on the index date, follow-up was divided into 3 periods: pre-diagnosis (-36 to -3 months, subdivided by year), peri-diagnosis (-3 to +3 months), and post-diagnosis (+3 to +12 months). Healthcare measures of interest were outpatient encounters, diagnoses of joint symptoms and conditions besides JIA (e.g., pain, stiffness, effusion, contracture, sprain, transient synovitis), number of dispensed medication classes, emergency department (ED) visits, and hospitalization. Dispensed medications of interest were nonsteroidal anti-inflammatory drugs (NSAIDs), glucocorticoids, opioids, and muscle relaxants. Median counts of each measure per person per period were compared between cases and matched controls using Wilcoxon signed-rank tests. Quarterly incidence rate ratios (IRRs) for measures in cases vs. controls were estimated and graphed using a negative binomial mixed-effects event-study model, which included a random intercept to account for matching and fixed effects for relative quarter and calendar quarter.
Results: We identified 2,867 eligible cases and 20,730 matched controls (Table). Median counts of total outpatient encounters, joint symptoms, and dispensed medication classes were significantly higher among cases than among controls across all periods (p< 0.001), rising steadily before diagnosis until peaking in the quarter just before (joint symptoms) or just after diagnosis (total outpatient encounters and medication classes) (Figure 1). Similarly, rates of ED visits and hospitalizations rose among cases relative to controls within 1-2 years before the index date and peaked peri-diagnosis (p< 0.001). Similar patterns were also noted for dispensed medications, although quarterly rates of dispensed opioids and muscle relaxants appeared to rise through one year post-diagnosis (Figure 2).
Conclusion: In a large low-income US population, high rates of encounters, joint symptoms, and medications in children with JIA up to 3 years before diagnosis strongly suggest diagnostic delays. Additionally, the persistent use of pain medications up to one year after diagnosis suggests sub-standard effectiveness of initiated therapies. Educational efforts should be directed both to the general pediatric community to enhance JIA recognition and to the rheumatology community to enhance early initiation of effective disease modifying therapies.
Table: Characteristics of eligible cases and matched controls.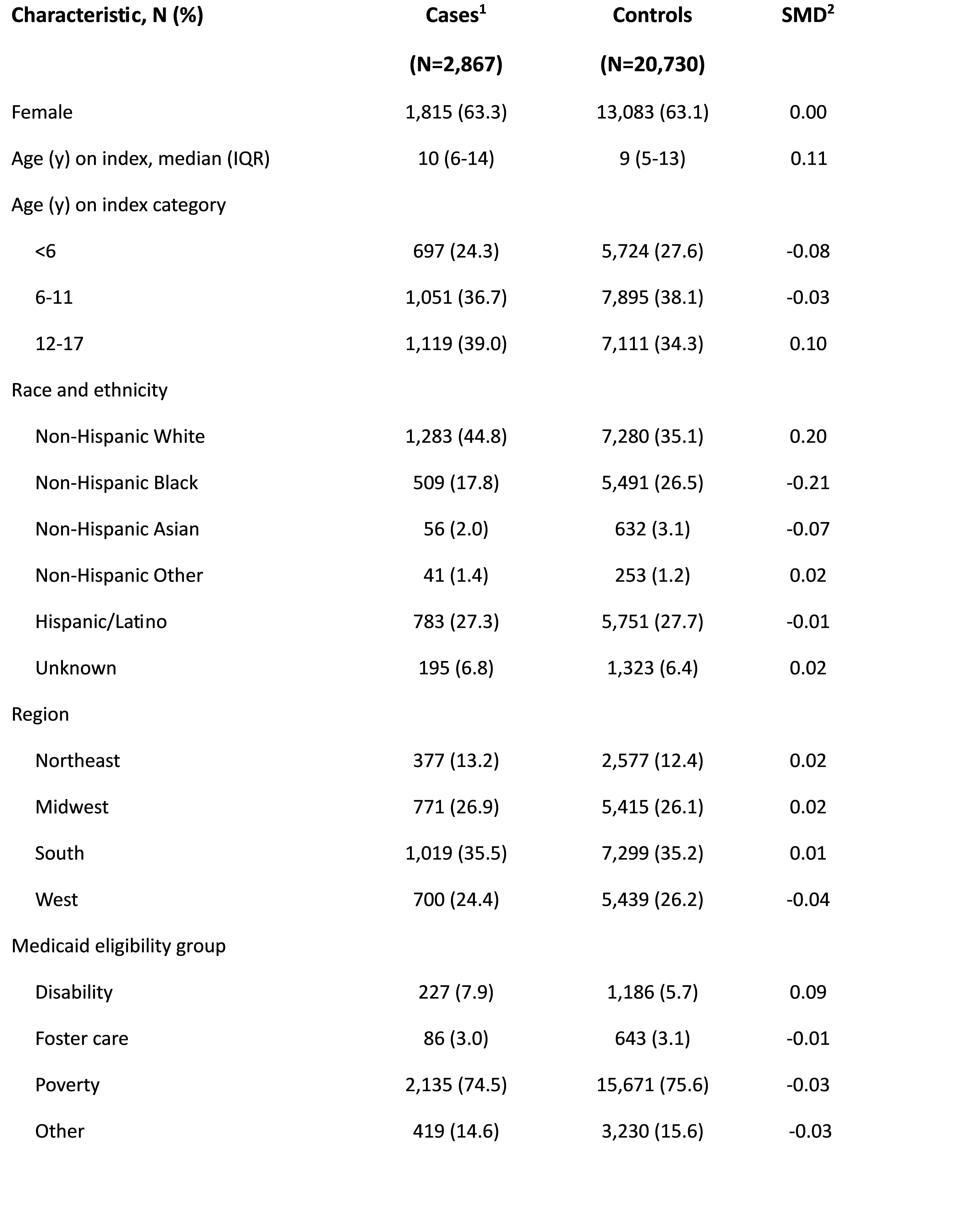 IP = inpatient; IQR = interquartile range; JIA = juvenile idiopathic arthritis; SMD = standardized mean difference.
IP = inpatient; IQR = interquartile range; JIA = juvenile idiopathic arthritis; SMD = standardized mean difference.
1, Cases were identified by ≥1 rheumatologist’s diagnosis plus ≥4 lab orders within ±4 months of index diagnosis, or ≥5 outpatient diagnoses that were 8-52 weeks apart, with the index diagnosis not from an ophthalmologist, plus any JIA treatment in 4 months post index diagnosis.
2, SMD with absolute value >0.1 reflects substantial covariate imbalance.
3, Birth inception cohort included children registered within 3 months after birth (96.6% within 1 week after birth).
Figure 1. Comparison of selected healthcare utilization measures between cases JIA and matched controls from 3 years before to 1 year after JIA diagnosis.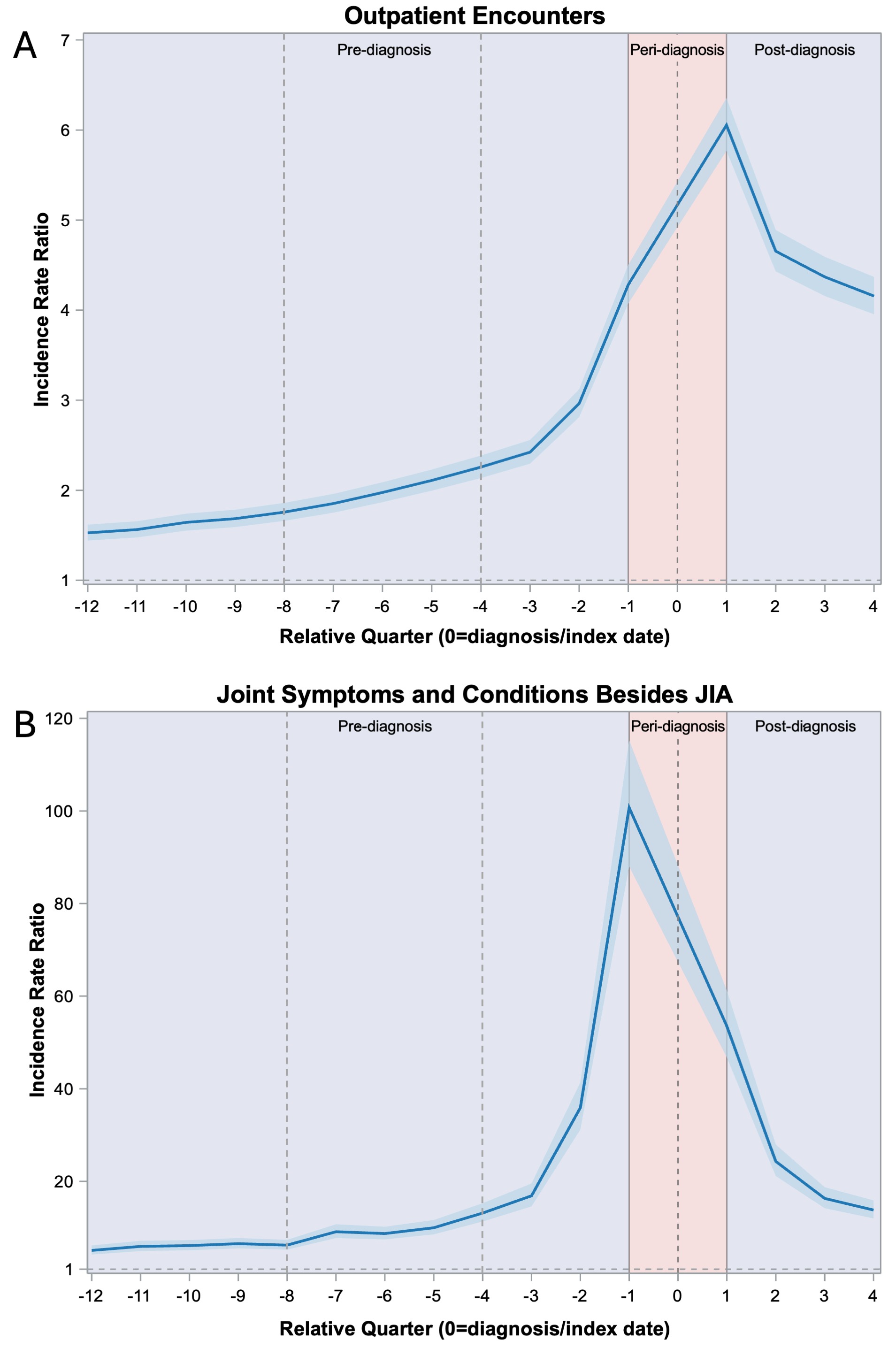 Figures illustrate model-adjusted rates of outpatient encounters (A) and encounters for joint symptoms or conditions besides JIA (B) based on International Classification of Diseases-9 or 10, Clinical Modification codes, in cases with JIA relative to matched controls. Quarterly incident rate ratios with 95% confidence interval bands in cases vs. matched controls were estimated using a negative binomial mixed-effects event-study model. Vertical dotted lines indicate yearly intervals.
Figures illustrate model-adjusted rates of outpatient encounters (A) and encounters for joint symptoms or conditions besides JIA (B) based on International Classification of Diseases-9 or 10, Clinical Modification codes, in cases with JIA relative to matched controls. Quarterly incident rate ratios with 95% confidence interval bands in cases vs. matched controls were estimated using a negative binomial mixed-effects event-study model. Vertical dotted lines indicate yearly intervals.
Figure 2. Comparison of selected dispensed medications between cases JIA and matched controls from 3 years before to 1 year after JIA diagnosis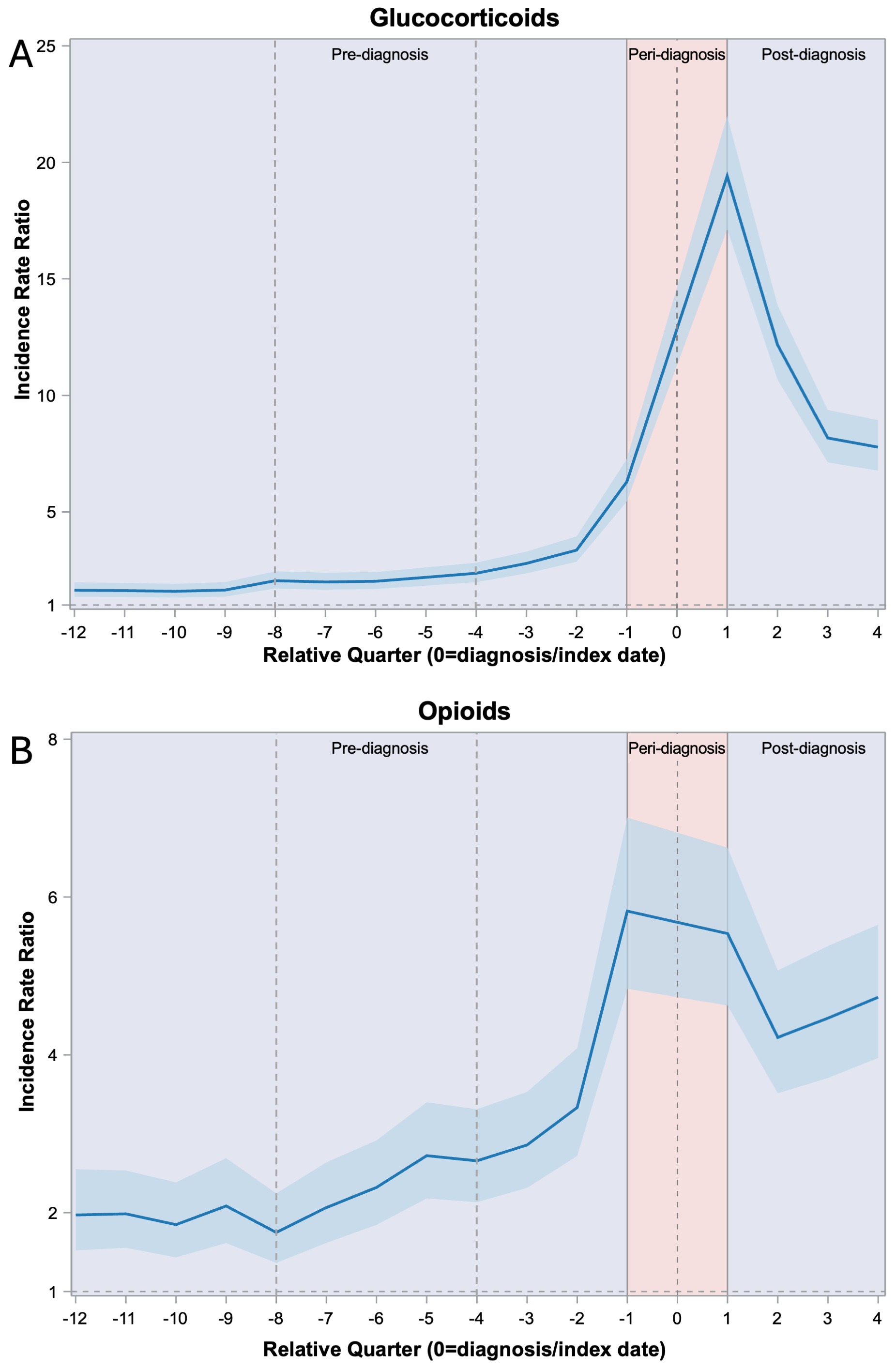 Figures illustrate model-adjusted rates of dispensed glucocorticoids (A) and opioids (B) in cases with JIA relative to matched controls. Quarterly incident rate ratios with 95% confidence interval bands in cases vs. matched controls were estimated using a negative binomial mixed-effects event-study model. Vertical dotted lines indicate yearly intervals.
Figures illustrate model-adjusted rates of dispensed glucocorticoids (A) and opioids (B) in cases with JIA relative to matched controls. Quarterly incident rate ratios with 95% confidence interval bands in cases vs. matched controls were estimated using a negative binomial mixed-effects event-study model. Vertical dotted lines indicate yearly intervals.
To cite this abstract in AMA style:
Mannion M, Zhu Y, Rege S, Varga J, Crystal S, Gerhard T, Rose C, Strom B, Horton D. Healthcare and Medication Utilization Before and After JIA diagnosis in Publicly Insured Children in the United States [abstract]. Arthritis Rheumatol. 2026; 78 (suppl 3). https://acrabstracts.org/abstract/healthcare-and-medication-utilization-before-and-after-jia-diagnosis-in-publicly-insured-children-in-the-united-states/. Accessed .« Back to 2026 Pediatric Rheumatology Symposium
ACR Meeting Abstracts - https://acrabstracts.org/abstract/healthcare-and-medication-utilization-before-and-after-jia-diagnosis-in-publicly-insured-children-in-the-united-states/
