Session Information
Session Time: 6:00PM-7:00PM
Background/Purpose: JDM is a rare heterogenous autoimmune condition. Pathogenesis is incompletely understood, though dysregulated type I/II IFN signaling is well-documented. Patients with refractory JDM had clinical improvement on systemic janus kinase inhibition (baricitinib) (Kim, 2021), which inhibits IFN signaling. This study aimed to assess cell-specific changes in IFN signaling with this treatment.
Methods: Peripheral blood mononuclear cells from 4 refractory JDM patients before and after baricitinib treatment (week 0, 4, 24, 72) were analyzed. All patients met the Bohan and Peter classification criteria for definite or probable JDM. Single-cell RNA sequencing (10X Genomics) data was processed using CellRanger (v7.1.0) software. Data was processed with Seurat (v4), with Azimuth (v0.4.5) cell type annotation. Cell types with <30 cells were excluded from analysis. Genes detectable in our data from three “IFN scores” (hallmark IFN α or type I IFN response (IRG α), hallmark IFN γ or type II IFN response (IRG γ), both from https://www.gsea-msigdb.org, and previously validated interferonopathy 28 IFN-regulated genes (IRG 28) (Kim, 2018)) were analyzed by Seurat module score (R) per cell, comparing patient-specific baseline to follow-up timepoints by Wilcoxon rank sum test with Bonferroni correction.
Results: The 3 IFN-response gene (IRG) lists and overlaps are shown in Figure 1. Comparing significant decreases in IFN scores overtime from the 4 JDM patients (Table 1), CD4 central memory T (TCM) cells had the most frequent significant decreases in IFN scores (97% overall, 12/12 IRG α, 11/12 IRG γ, 12/12 IRG 28), followed by CD8 naïve cells (94% overall, 11/12 IRG α, 12/12 IRG γ, 11/12 IRG 28). Mucosal-Associated Invariant T (MAIT) cells (64% overall, 8/11 IRG α, 9/11 IRG γ, 4/11 IRG-28) and CD14 monocytes (78% overall, 8/9 IRG α, 8/9 IRG γ, 5/9 IRG-28) had the least frequent significant decreases in IFN scores. By patient, significant IFN score decreases across cell types and timepoints ranged from 40/54 (74%) to 39/42 (93%). By timepoint, significant decreases in IFN score across patients and cell types ranged from 83% (19/22 IRG α, 8/22 IRG γ, 18/22 IRG 28) at timepoint 4 (T4, 72 weeks) to 86% (18/22 IRG α, 22/22 IRG γ, 17/22 IRG 28) at T3 (24 weeks). The IRG γ score had the most frequent significant decreases (58/65, 89%), followed by IRG α (56/65, 86%), and IRG 28 (52/65, 80%). One patient had significant increases in score at T4 CD14 monocytes (IRG α, IRG γ) and B cells (IRG γ).
Conclusion: Refractory JDM patients had decreases in 3 IRG scores (α, γ, 28) in most peripheral cell types at most timepoints (4-72 weeks) on baricitinib. CD4 TCM cells were the most sensitive to baricitinib (most frequent significant decreases in IFN scores), while MAIT cells were the least sensitive (least frequent significant decreases in IFN scores). IRG γ score was the most sensitive to baricitinib while IRG 28 score was the least. Type I and II IFN regulated gene scores decreased in multiple cell types on baricitinib, correlating with clinical response.
Acknowledgments: NIH IRP, NIAMS including Genomic Technology Section, NIEHS.
Table 1. Features of JDM patients at baseline.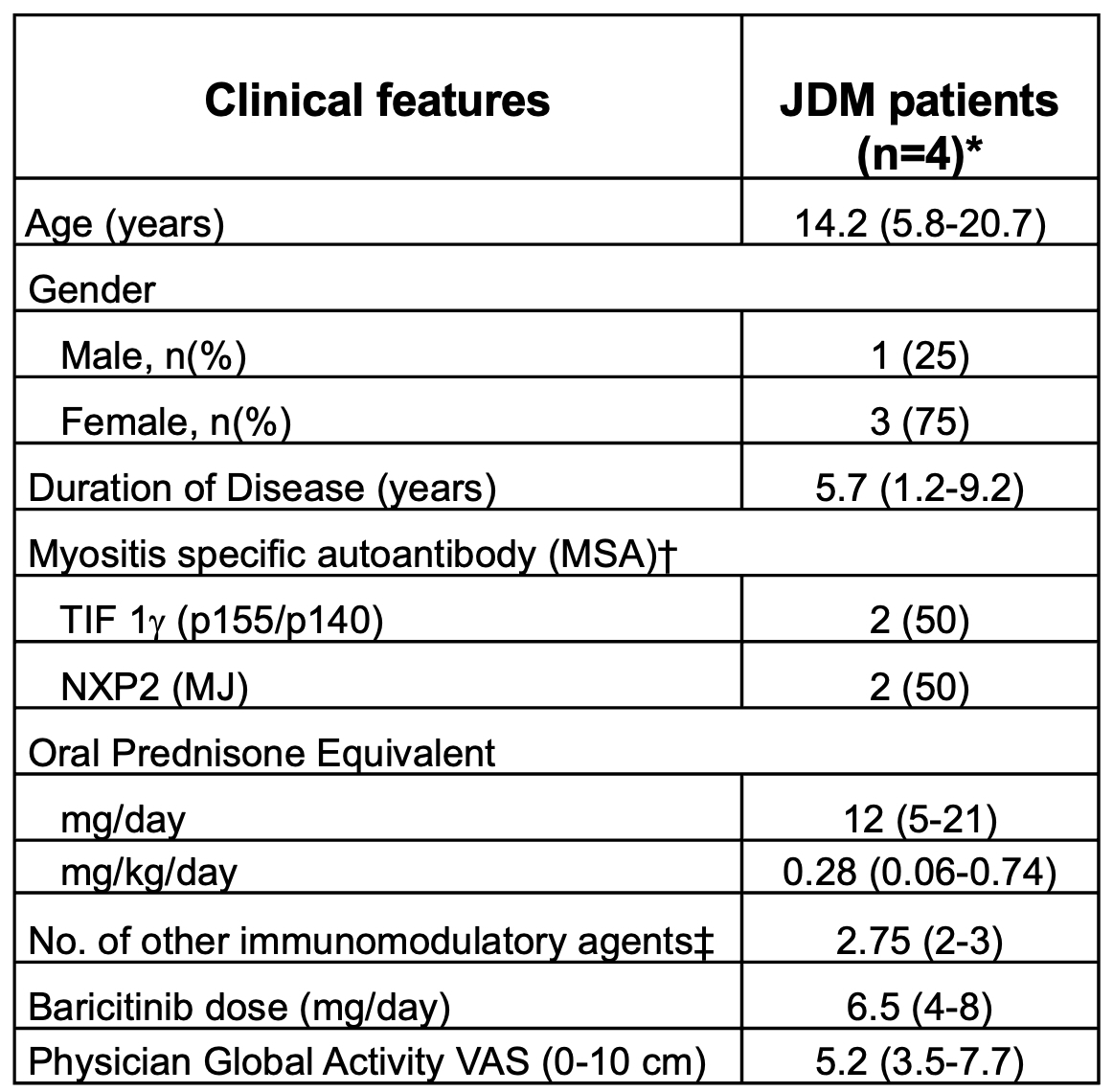 *n (%) or mean (range)
*n (%) or mean (range)
†Performed at Oklahoma Medical Research Foundation (OMRF) laboratory. ‡Intravenous immunoglobulin (n=4), methotrexate (n=1), mycophenolate mofetil (MMF) (n=4), cyclosporin (n=1), tacrolimus (n=1). TIF 1γ: transcriptional intermediary factor 1γ; NXP2: nuclear matrix protein 2; VAS: visual analog scale.
Some information adapted from Kim H et al, Ann Rheum Dis. 2021
Figure 1.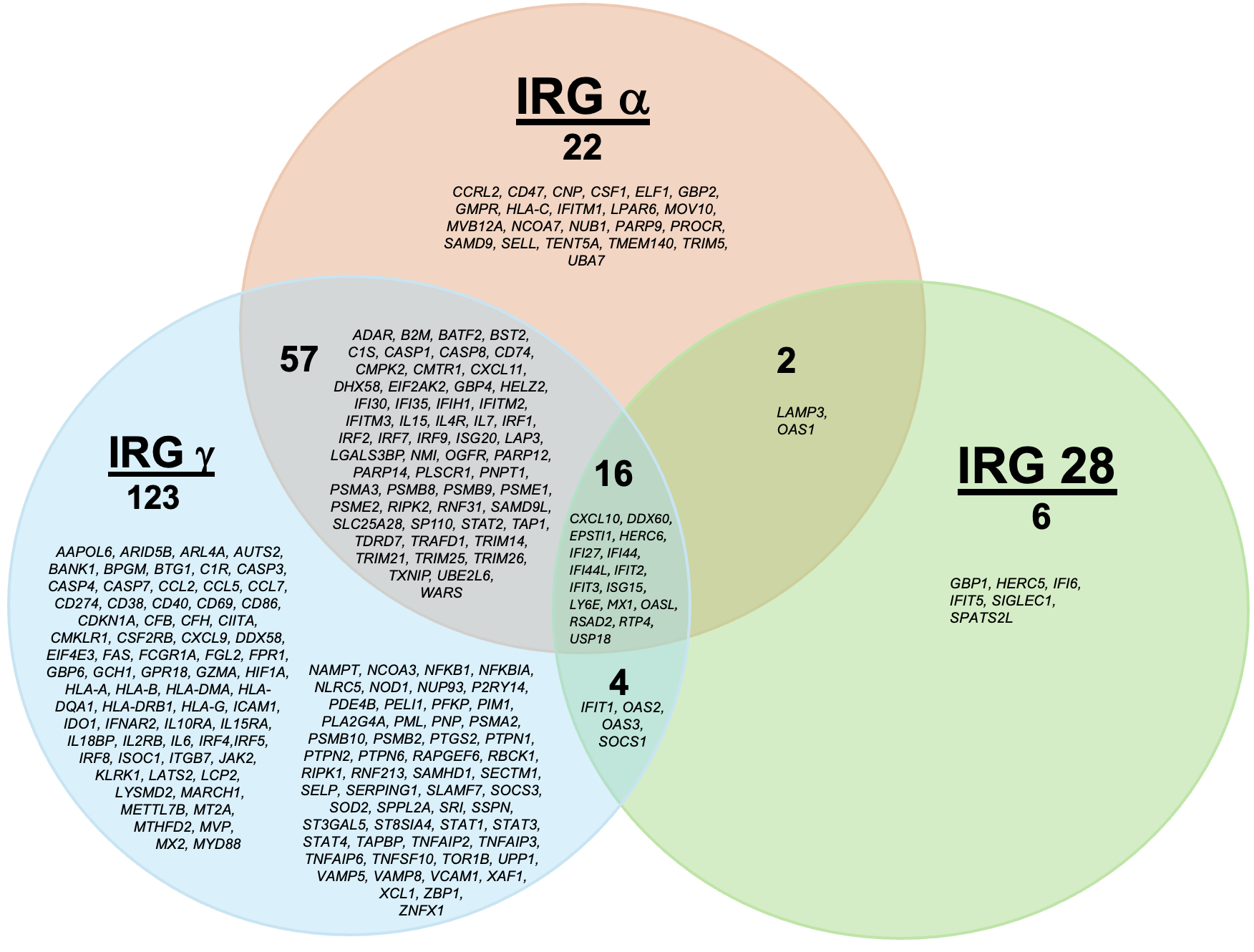 Venn diagram of Interferon gene lists.
Venn diagram of Interferon gene lists.
Interferon alpha (IRG α) and interferon gamma (IRG γ) gene lists are from the Molecular Signatures Database lists (https://www.gsea-msigdb.org). 28-gene IFN-regulated gene list (IRG 28) is from Kim et al, J Interferon Cytokine Res. 2018. Genes are listed alphabetically.
To cite this abstract in AMA style:
Simpson J, Chen V, Chin A, Zajmi U, Kaneshiro A, Darrell M, Brooks S, Colbert R, Rider L, Dell’Orso S, Naz F, Kim H. Longitudinal Single Cell Transcriptomic Analysis of Type I and II Interferon (IFN) Response Genes (IRG) in Refractory Juvenile Dermatomyositis (JDM) Patients with Baricitinib Treatment [abstract]. Arthritis Rheumatol. 2026; 78 (suppl 3). https://acrabstracts.org/abstract/longitudinal-single-cell-transcriptomic-analysis-of-type-i-and-ii-interferon-ifn-response-genes-irg-in-refractory-juvenile-dermatomyositis-jdm-patients-with-baricitinib-treatment/. Accessed .« Back to 2026 Pediatric Rheumatology Symposium
ACR Meeting Abstracts - https://acrabstracts.org/abstract/longitudinal-single-cell-transcriptomic-analysis-of-type-i-and-ii-interferon-ifn-response-genes-irg-in-refractory-juvenile-dermatomyositis-jdm-patients-with-baricitinib-treatment/
