Session Information
Session Time: 5:17PM-5:22PM
Background/Purpose: MAS is a life-threatening complication of SLE, characterized by interferon-gamma (IFNg)-driven macrophage activation and systemic hyperinflammation. There is substantial preclinical evidence of IFNg having a role in MAS in SLE, but there is limited clinical data to confirm these observations. Emapalumab, an anti-IFNg antibody, binds free and receptor-bound IFNg, providing rapid and targeted neutralization of IFNg. Emapalumab has previously demonstrated sustained control of MAS in patients (pts) with Still’s disease. This report presents the first clinical trial data from pts with MAS in SLE administered emapalumab
Methods: Cohort 2 of Study NI-0501-14 (EMERALD; NCT05001737) investigated the efficacy and safety of emapalumab in pts with MAS in SLE who had an inadequate response to high-dose glucocorticoids (GCs). Pts were planned to be treated with emapalumab for 4 weeks: a 6 mg/kg loading dose, followed by 3 mg/kg every 3 days from days 4–16, then 3 mg/kg twice weekly until Day 28 (or longer if insufficient clinical response). The primary efficacy endpoint was complete response (CR) at Week 8, defined as resolution of clinical signs according to investigator assessment (visual analog scale [VAS] ≤1/10 cm) and normalization of 7 MAS-related laboratory parameters). Overall response (OR) was defined as CR or a partial response (VAS < 4/10 cm and normalization of ≥3 MAS-related laboratory parameters). Other endpoints included GC tapering, survival, biomarkers, and safety.
Results: Eight pts with an inadequate response to high-dose GCs were enrolled (5 [67.5%] females), with a median age of 16 years (range, 2–24; Table 1). All pts had lung and/or hepatic involvement. Six (75%) pts received 3–10 doses of emapalumab and treatment was administered for 1–< 4 weeks in 5 (63%) pts. Seven (88%) pts completed the study. One pt discontinued because of an adverse event (progressive decreased prothrombin activity). At Week 8, 4 (50%) pts achieved a CR with a median time to first CR of 5.9 weeks. Seven (88%) pts achieved a CR at any time. Six (75%) pts achieved an OR at Week 8 with a median time to first OR of 3.7 weeks. No pt had a recurrence of MAS during 1 year of follow-up. Mean GC dose reduced by 93% from 9.34 mg/kg/day at baseline to 0.65 mg/kg/day at Week 8 (Table 2). Median time to any GC tapering (50% reduction from baseline or reduction to dose prior to MAS episode) was 1.0 week. Median time to weekly average daily GC dose of ≤1 mg prednisolone-equivalent/kg/day was 5.5 weeks. No new safety concerns were identified. Four serious adverse drug reactions were reported in 4 pts. No infusion-related reactions occurred. No deaths occurred.
Conclusion: Emapalumab rapidly controlled MAS in SLE in 75% of pts with an initial inadequate response to high-dose GC treatment administered emapalumab. This suggests that IFNg is a pivotal cytokine in pts with MAS in SLE and similar outcomes can be achieved for patients with MAS treated with emapalumab, independently of the underlying rheumatic disease (Still’s disease or SLE).
Table 1: Demographics, baseline characteristics and treatment outcomes for in patients with macrophage activation syndrome in systemic lupus erythematosus administered emapalumab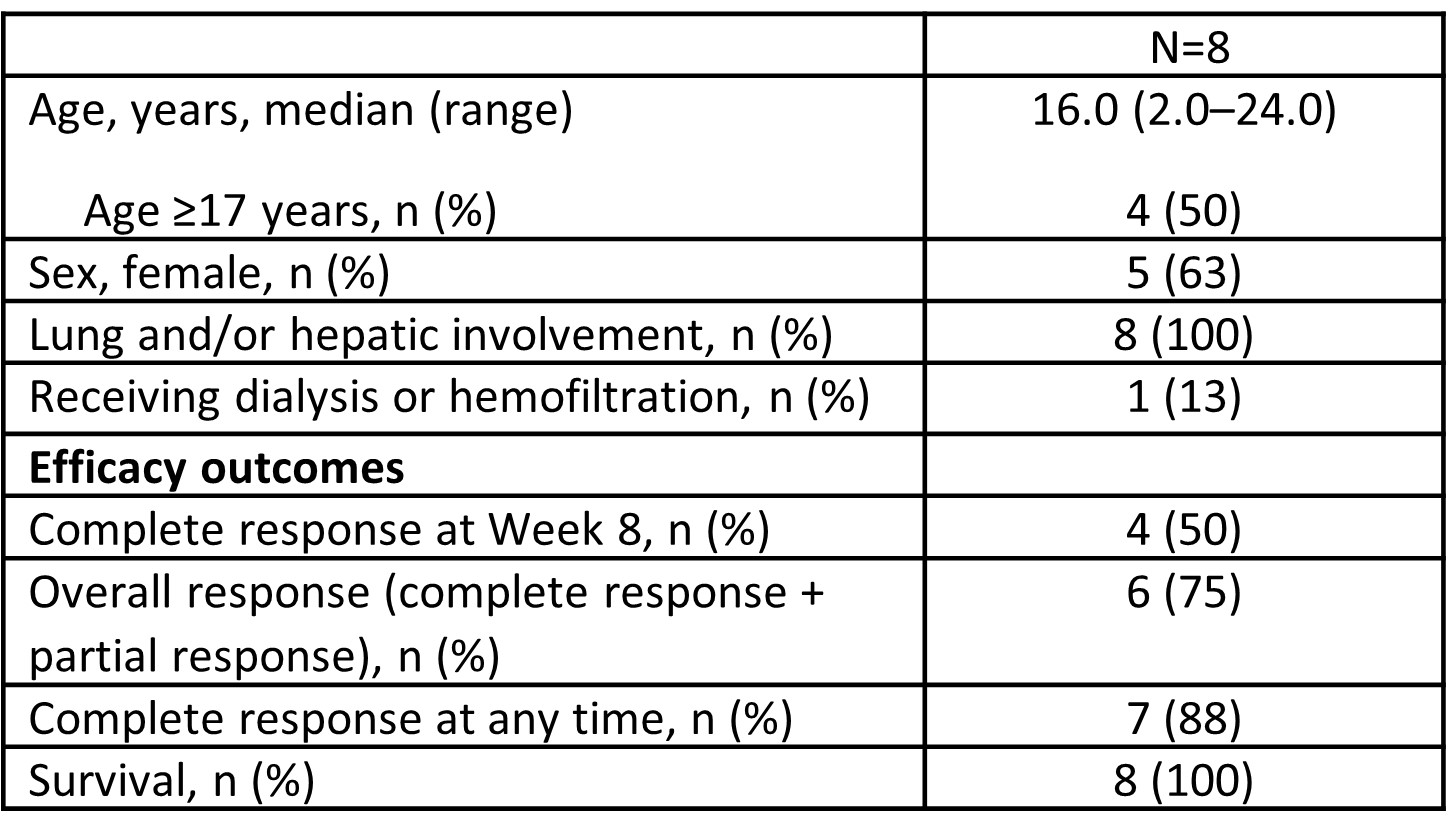
Table 2: Glucocorticoid dosing in patients with macrophage activation syndrome in systemic lupus erythematosus administered emapalumab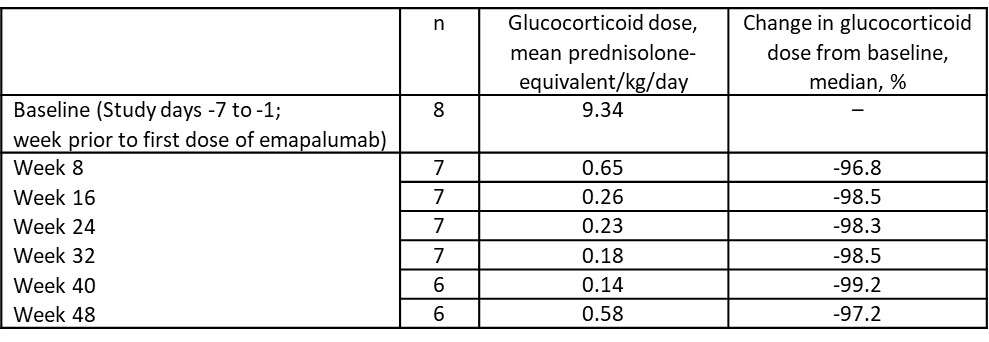
To cite this abstract in AMA style:
Grom A, Akoghlanian S, elder m, Modica R, Mallett S, Jamieson B, De Benedetti F. Efficacy and Safety of Emapalumab in Patients with Macrophage Activation Syndrome (MAS) in Systemic Lupus Erythematosus (SLE) [abstract]. Arthritis Rheumatol. 2026; 78 (suppl 3). https://acrabstracts.org/abstract/efficacy-and-safety-of-emapalumab-in-patients-with-macrophage-activation-syndrome-mas-in-systemic-lupus-erythematosus-sle/. Accessed .« Back to 2026 Pediatric Rheumatology Symposium
ACR Meeting Abstracts - https://acrabstracts.org/abstract/efficacy-and-safety-of-emapalumab-in-patients-with-macrophage-activation-syndrome-mas-in-systemic-lupus-erythematosus-sle/
