Session Information
Date: Thursday, March 19, 2026
Title: Plenary Abstract Session I
Session Time: 3:00PM-3:15PM
Background/Purpose: Juvenile systemic scleroderma (jSSc) is a rare autoimmune disease characterized by progressive fibrosis and vascular dysfunction. Although jSSc shares important clinical features with adult SSc, including interstitial lung disease in ~40% of patients, reflecting substantial fibrotic burden, its molecular drivers remain poorly defined. In contrast to adult SSc, where multiple single-cell studies have mapped fibroblast heterogeneity, single-cell analyses in jSSc are extremely limited; our group generated the first dataset (n=3 paired, presented in prior meetings). Here we present the largest single-cell dataset to date in jSSc (n=9) and map macrophage–fibroblast communication networks that sustain chronic fibrosis.
Methods: Skin biopsies from 9 jSSc patients and 9 age-matched healthy controls underwent scRNA-seq using the 10x Genomics® Chromium platform. Processed single-cell suspensions were analyzed using Seurat and Harmony for normalization, clustering, and batch correction, with UMAP visualization. Cell-to-cell communication was assessed using NicheNet and CellChat to predict macrophage–fibroblast ligand–receptor interactions and downstream target gene programs.
Results: Eleven transcriptionally distinct fibroblast subsets were identified. COL11A1⁺/POSTN⁺ and a novel STC1⁺/FZD9⁺ fibroblast population were markedly expanded in jSSc and enriched for TGFβ, WNT, and FGF signaling driving extracellular matrix accumulation. The STC1⁺/FZD9⁺ subset showed strong enrichment of pulmonary and hepatic fibrosis signaling pathways. Macrophage clusters MMP19⁺/CTSL⁺ and MERTK⁺ emerged as key profibrotic “sender” populations expressing ligands TGFB1, AREG, and COL6A2, signaling to fibroblast receptors TGFBR1/2, FGFR1/2, and FZD9. NicheNet and CellChat cell communication software revealed an integrated macrophage–fibroblast communication network incorporating TGFβ, FGF, WNT, IGF, NOTCH, and TNF pathways. Accessory activators (ITGAV, THBS1, SDC2) amplified TGFβ engagement, suggesting a self-reinforcing fibroblast-activation loop independent of additional inflammatory input.
Conclusion: This study provides the first expanded single-cell atlas of jSSc skin and identifies a coordinated macrophage-driven signaling circuit sustaining profibrotic fibroblast states. While major pathways parallel adult SSc, the fibroblast populations involved, particularly STC1⁺/FZD9⁺ cells, appear distinct to jSSc. TGFβ–FGF–WNT pathway convergence and accessory activation through ITGAV, THBS1, and SDC2 highlight potential therapeutic targets. By defining juvenile-specific immune–stromal communication, this work advances mechanistic understanding of fibrosis propagation in jSSc and identifies candidate nodes for intervention.
Figure 1. Single-cell characterization of fibroblasts and macrophages in juvenile systemic scleroderma (jSSc) skin.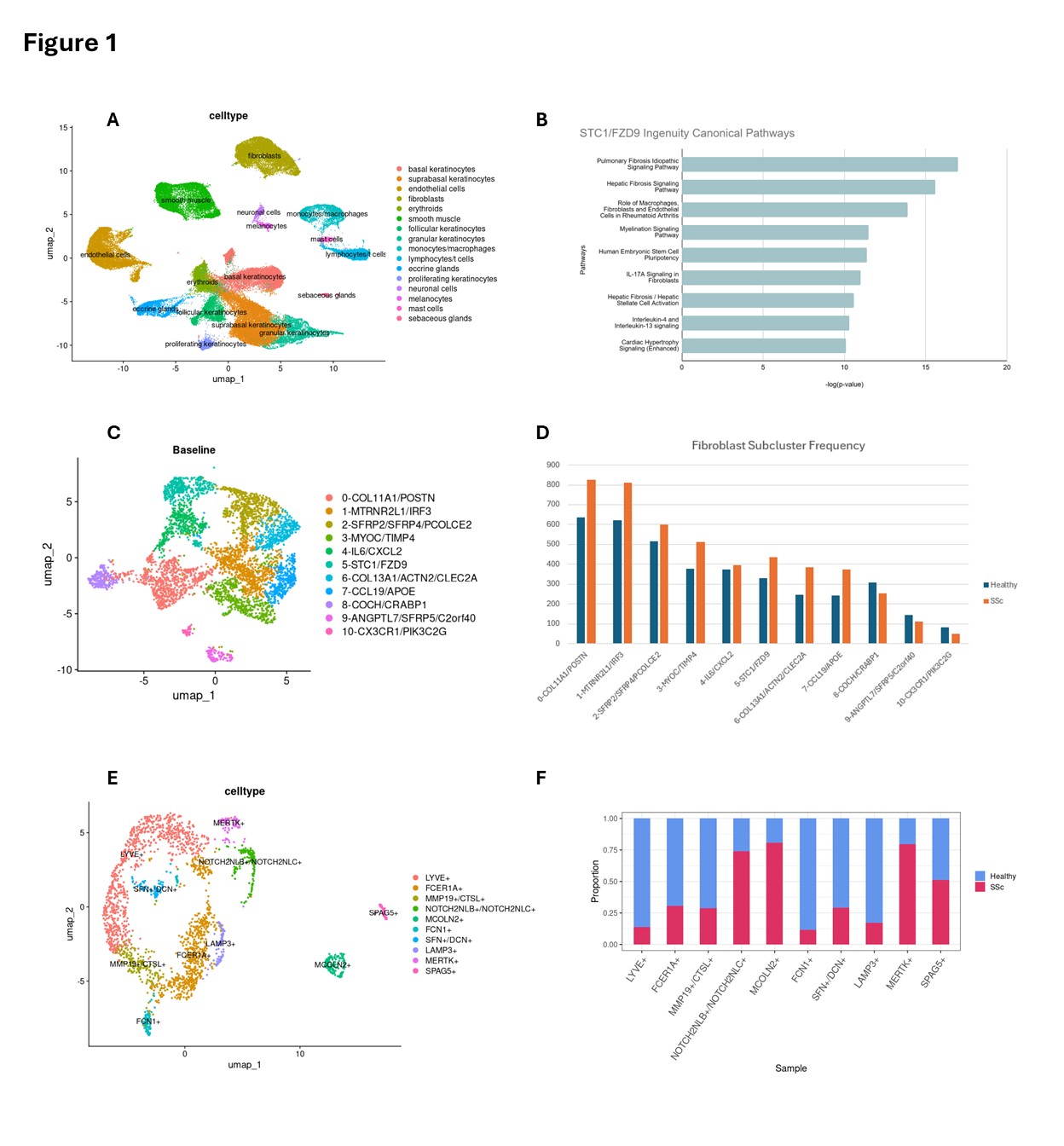 Fig 1. (A) UMAP plot showing cell-type annotations across all jSSc and healthy skin samples, highlighting major populations including fibroblasts, macrophages, T cells, and endothelial cells. (B) Ingenuity Pathway Analysis of the novel STC1⁺/FZD9⁺ fibroblast cluster demonstrating enrichment of profibrotic pathways, including pulmonary and hepatic fibrosis signaling and TGFβ pathway activation. (C) UMAP of fibroblast subclusters identifying 11 transcriptionally distinct fibroblast subsets based on marker gene expression. (D) Bar plot comparing fibroblast subcluster frequencies between healthy and jSSc samples, showing marked expansion of COL11A1⁺/POSTN⁺ and STC1⁺/FZD9⁺ fibroblasts in jSSc skin. (E) UMAP of macrophage subtypes annotated by key markers, revealing profibrotic subsets including MMP19⁺/CTSL⁺ and MERTK⁺ macrophages. (F) Proportion of macrophage subtypes in healthy versus jSSc skin, demonstrating enrichment of profibrotic macrophages in disease samples.
Fig 1. (A) UMAP plot showing cell-type annotations across all jSSc and healthy skin samples, highlighting major populations including fibroblasts, macrophages, T cells, and endothelial cells. (B) Ingenuity Pathway Analysis of the novel STC1⁺/FZD9⁺ fibroblast cluster demonstrating enrichment of profibrotic pathways, including pulmonary and hepatic fibrosis signaling and TGFβ pathway activation. (C) UMAP of fibroblast subclusters identifying 11 transcriptionally distinct fibroblast subsets based on marker gene expression. (D) Bar plot comparing fibroblast subcluster frequencies between healthy and jSSc samples, showing marked expansion of COL11A1⁺/POSTN⁺ and STC1⁺/FZD9⁺ fibroblasts in jSSc skin. (E) UMAP of macrophage subtypes annotated by key markers, revealing profibrotic subsets including MMP19⁺/CTSL⁺ and MERTK⁺ macrophages. (F) Proportion of macrophage subtypes in healthy versus jSSc skin, demonstrating enrichment of profibrotic macrophages in disease samples.
Figure 2. Macrophage–fibroblast communication networks driving fibrosis in juvenile systemic scleroderma.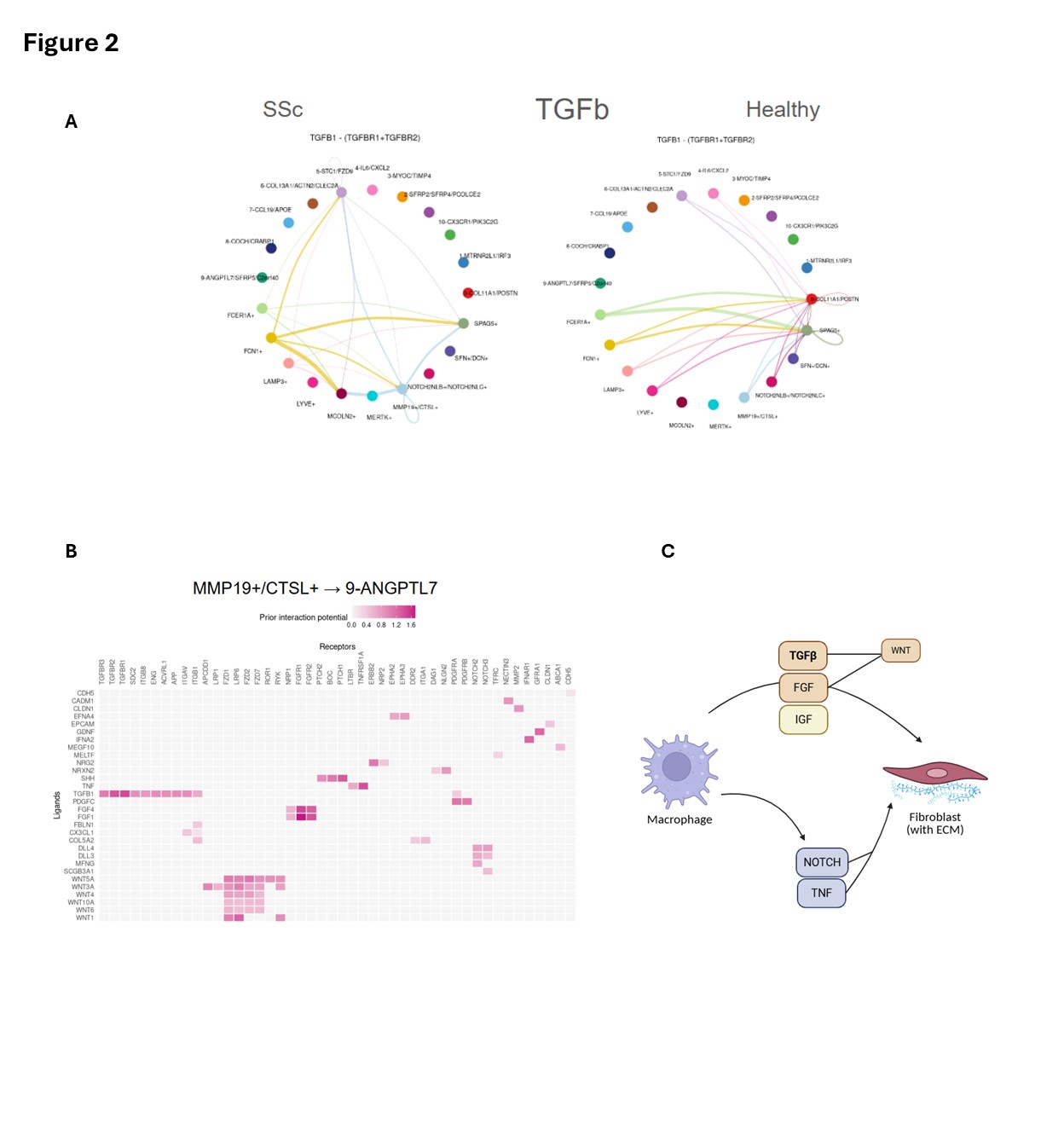 Fig 2. (A) CellChat analysis of TGFβ ligand–receptor signaling between macrophage and fibroblast subtypes in jSSc (left) versus healthy (right) skin, demonstrating broader and stronger intercluster connectivity in jSSc. (B) NicheNet ligand–receptor interaction heatmap highlighting strong signaling potential from MMP19⁺/CTSL⁺ macrophages to ANGPTL7⁺ fibroblasts via ligands TGFB1, AREG, and COL6A2 and receptors TGFBR1, FGFR1, and FZD9. (C) Schematic model summarizing the macrophage-driven profibrotic circuit: macrophage-derived TGFβ, FGF, and IGF activate fibroblast ECM programs via WNT signaling, while NOTCH and TNF pathways reinforce chronic fibroblast activation. Accessory activators (ITGAV, THBS1, SDC2) sustain this signaling loop independent of additional inflammatory cues.
Fig 2. (A) CellChat analysis of TGFβ ligand–receptor signaling between macrophage and fibroblast subtypes in jSSc (left) versus healthy (right) skin, demonstrating broader and stronger intercluster connectivity in jSSc. (B) NicheNet ligand–receptor interaction heatmap highlighting strong signaling potential from MMP19⁺/CTSL⁺ macrophages to ANGPTL7⁺ fibroblasts via ligands TGFB1, AREG, and COL6A2 and receptors TGFBR1, FGFR1, and FZD9. (C) Schematic model summarizing the macrophage-driven profibrotic circuit: macrophage-derived TGFβ, FGF, and IGF activate fibroblast ECM programs via WNT signaling, while NOTCH and TNF pathways reinforce chronic fibroblast activation. Accessory activators (ITGAV, THBS1, SDC2) sustain this signaling loop independent of additional inflammatory cues.
To cite this abstract in AMA style:
Shethji A, Hutchins T, Sanyal A, Torok K. Dysregulated Macrophage–Fibroblast Crosstalk Identified by Single-Cell RNA-seq in Juvenile Systemic Scleroderma Skin [abstract]. Arthritis Rheumatol. 2026; 78 (suppl 3). https://acrabstracts.org/abstract/dysregulated-macrophage-fibroblast-crosstalk-identified-by-single-cell-rna-seq-in-juvenile-systemic-scleroderma-skin/. Accessed .« Back to 2026 Pediatric Rheumatology Symposium
ACR Meeting Abstracts - https://acrabstracts.org/abstract/dysregulated-macrophage-fibroblast-crosstalk-identified-by-single-cell-rna-seq-in-juvenile-systemic-scleroderma-skin/
